[ad_1]
Sham surgery trials have shown that some of our most popular surgeries are themselves shams.
Intragastric balloons “arrived with much fanfare in the 1980s,” since they could be implanted into the stomach and inflated with air or water to fill much of the space. Unfortunately, surgical devices are often brought to the market before there is adequate evidence of effectiveness and safety, and the balloons were no exception.
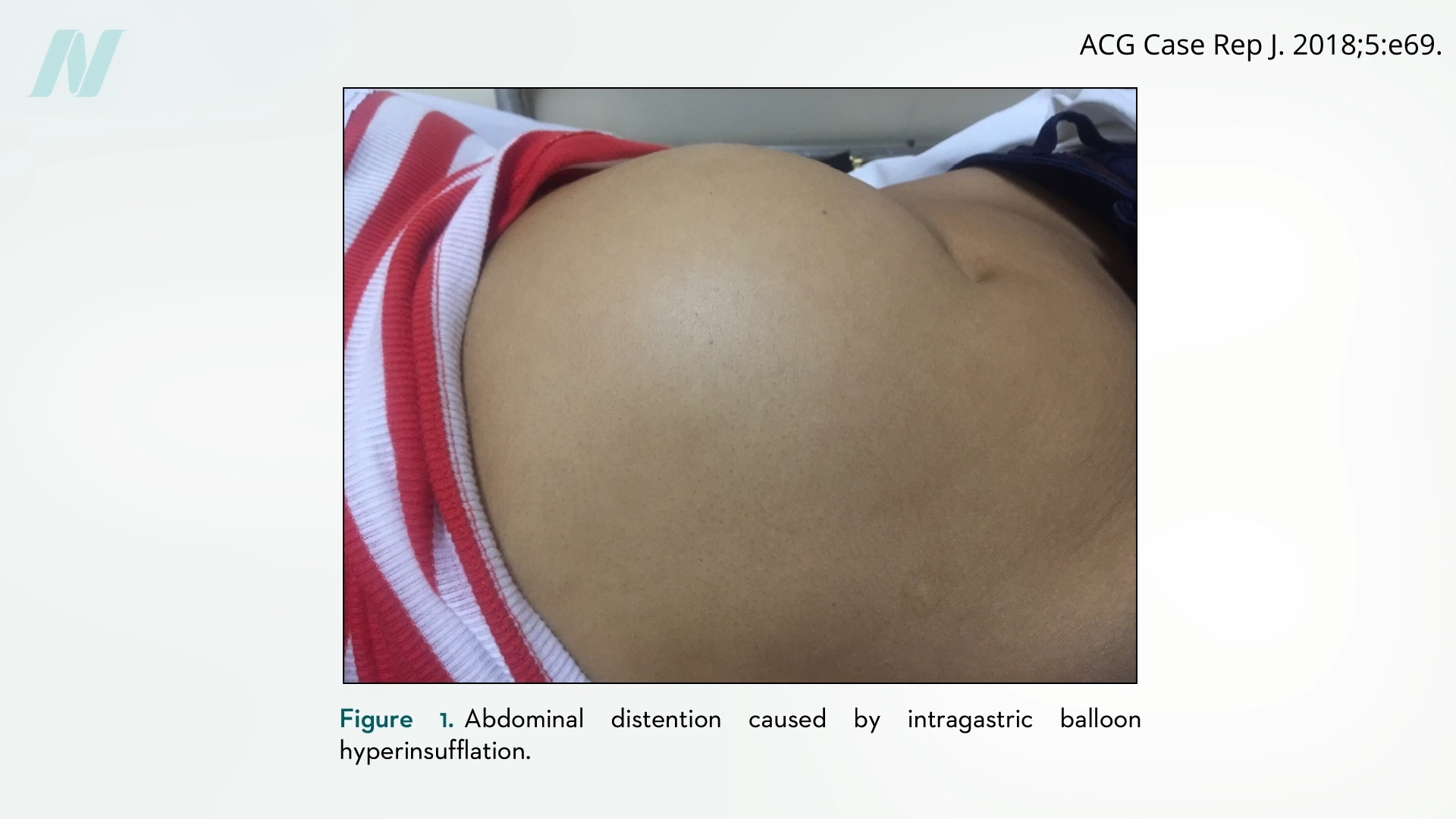
The “gastric bubble” had its bubble burst when a study at the Mayo Clinic found that 8 out of 10 balloons “spontaneously deflated,” which is potentially dangerous because they could pass into the intestines and cause an obstruction, as you can see below and at 0:40 in my video Is Gastric Balloon Surgery Safe and Effective for Weight Loss?.
Before balloons deflated, however, they apparently caused gastric erosions in half the patients, damaging their stomach lining. The kicker is that, in terms of inducing weight loss, they didn’t even work when compared to diet and other behavior modification strategies. Eventually, intragastric balloons were pulled from the market. But now, balloons are back.
After a 33-year hiatus, the U.S. Food and Drug Administration started approving a new slew of intragastric balloons in 2015, which immediately resulted in the placement of more than 5,000 devices. By then, the Sunshine Act had passed. It forced drug companies and the surgical and medical device industry to disclose any payments made to physicians, shining a disinfecting light on industry enticements. By now, most people know about the overly cozy financial relationships doctors can have with Big Pharma, but fewer may realize that surgeons can also get payments from the companies for the devices they use. The 100 top physician recipients of industry payments received an unbelievable $12 million from device companies in a single year. Yet outrageously, when they published papers, only a minority disclosed the blatant conflict of interest.
The benefit of balloons over most types of bariatric surgery is that they’re reversible, but that doesn’t mean they’re benign. The FDA has released a series of advisories about the risks, which include death. But how could someone suffer a stomach perforation with a smooth, rounded object? By that smooth, rounded object causing the patient to vomit so much that they rupture their stomach and die. Nausea and vomiting are unsurprisingly “very common side effects,” affecting the majority of those who have balloons placed inside of them. Persistent vomiting likely also explains cases of life-threatening nutrient deficiencies after balloon implantation.
Some complications, such as bowel obstruction, are due to the balloons deflating, but others, oddly enough, are due to the balloons suddenly overinflating, causing pain, vomiting, and abdominal distention, as you can see below and at 2:45 in my video.
This issue was first noticed in breast implants, as documented in reports such as “The Phenomenon of the Spontaneously Autoinflating Breast Implant.” Out of nowhere, the implants can just start growing, increasing breast volume by an average of more than 50%. “It remains an underreported and poorly understood phenomenon,” one review noted. (Interestingly, breast implants were actually used as some of the first failed experimental intragastric balloons.)
As with any medical decision, though, it’s all about risks versus benefits. Industry-funded trials display “notable weight loss,” but it’s hard to tease out the effect of the balloon on its own from the accompanying “supervised diet and lifestyle changes” prescribed in the studies. In drug trials, you can randomize study participants to sugar pills, but how do you eliminate the placebo effect of undergoing a procedure? Perform sham surgery.
In 2002, a courageous study was published in The New England Journal of Medicine. The most common orthopedic surgery—arthroscopic surgery of the knee—was put to the test. Billions of dollars are spent on sticking scopes into knee joints and cutting away damaged tissue in osteoarthritis and knee injuries, but does that actually work? People suffering from knee pain were randomized to get the actual surgery versus a sham surgery, in which surgeons sliced into people’s knees and pretended to perform the procedure—even splashing saline—without actually treating the joint.
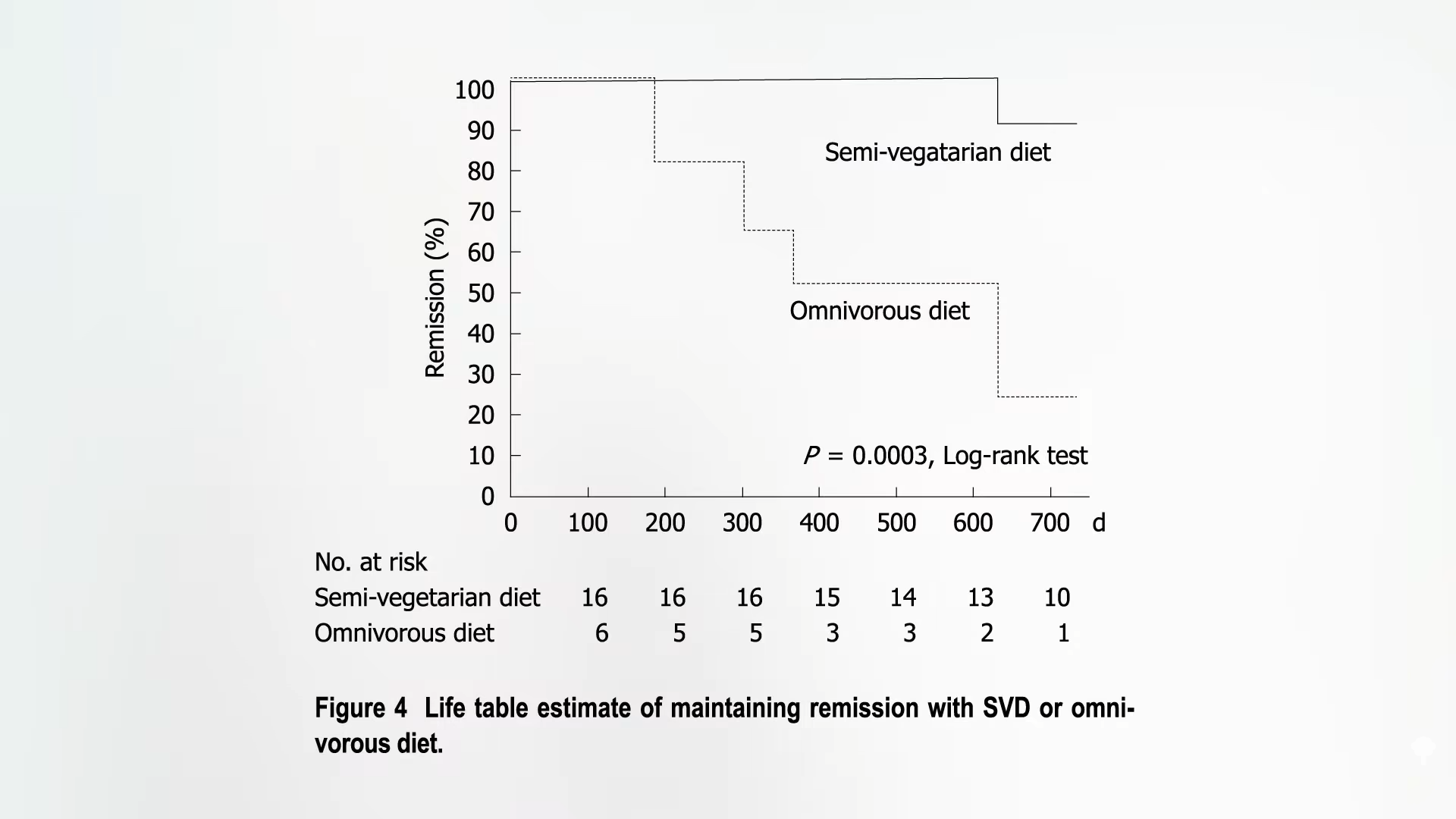
The trial caused an uproar. How could anyone randomize people to get cut open for fake surgery? Professional medical associations questioned the ethics of the surgeons as well as “the sanity” of the patients who agreed to be part of the trial. Guess what happened? The surgical patients got better, but so did the placebo patients, as you can see below and at 4:42 in my video.
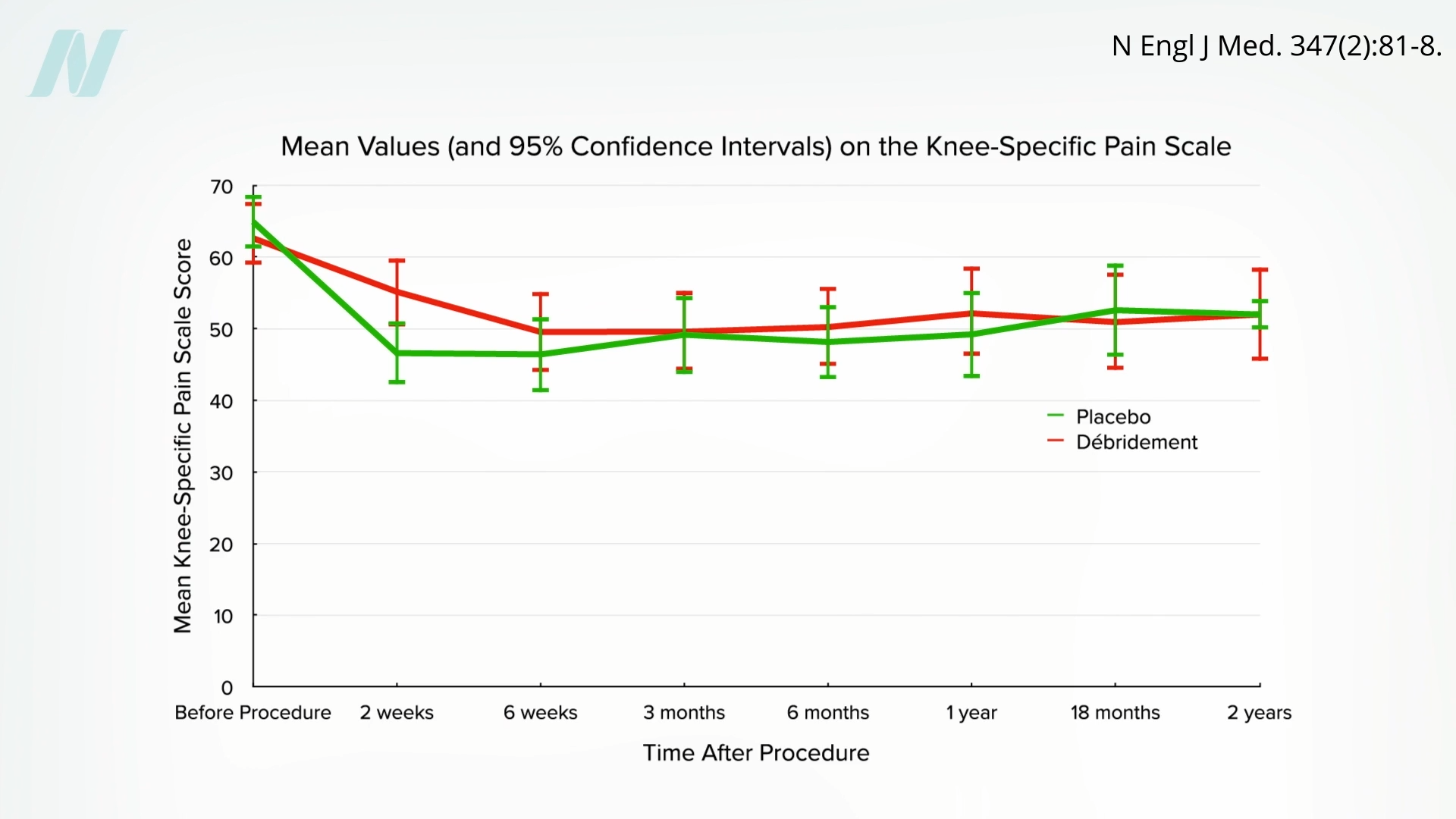
The surgeries had no actual effect. Currently, rotator cuff shoulder surgery is facing the same crisis of confidence.
When intragastric balloons were put to the test, sham-controlled trials showed that both older and newer devices sometimes fail to offer any weight-loss benefit. Even when they do work, the weight loss may be temporary because balloons are only allowed to stay in for six months (at which point the deflation risk gets too great). Why can’t you keep putting new ones in? That’s been tried; it failed to improve long-term weight outcomes. A sham-controlled trial showed that any effects of the balloon on appetite and satiety may vanish with time, perhaps as your body gets used to the new normal.
What sham surgery trials have shown us is that some of our most popular surgeries are themselves shams. Doctors like to pride themselves on being men and women of science. For example, we rightly rail against the anti-vaccination movement. Many of us in medicine have been troubled by the political trend in which people “choose their own facts.” But when I read that some of these still-popular surgeries are not only useless but may actually make matters worse (for example, increasing the risk of progression to a total knee replacement), I can’t help but think we are hardly immune to our own versions of fake news and alternative facts.
Doctor’s Note
Next in this two-part series is Extreme Weight-Loss Devices.
For more on bariatric surgery, check out related posts below.
My book How Not to Diet is focused exclusively on sustainable weight loss. Borrow it from your local library or pick up a copy from your favorite bookseller. (All proceeds from my books are donated to charity.)
[ad_2]
Michael Greger M.D. FACLM
Source link

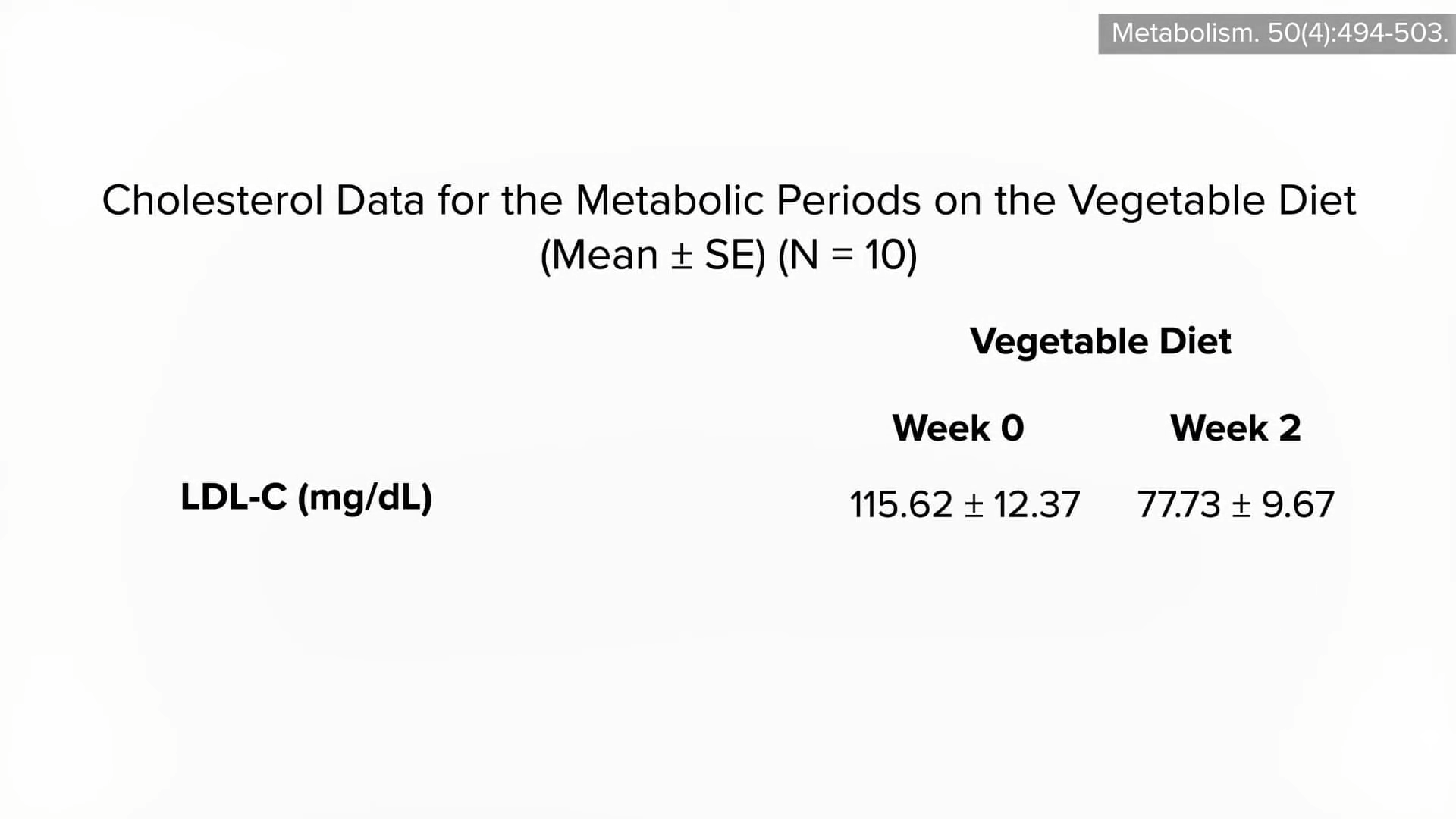

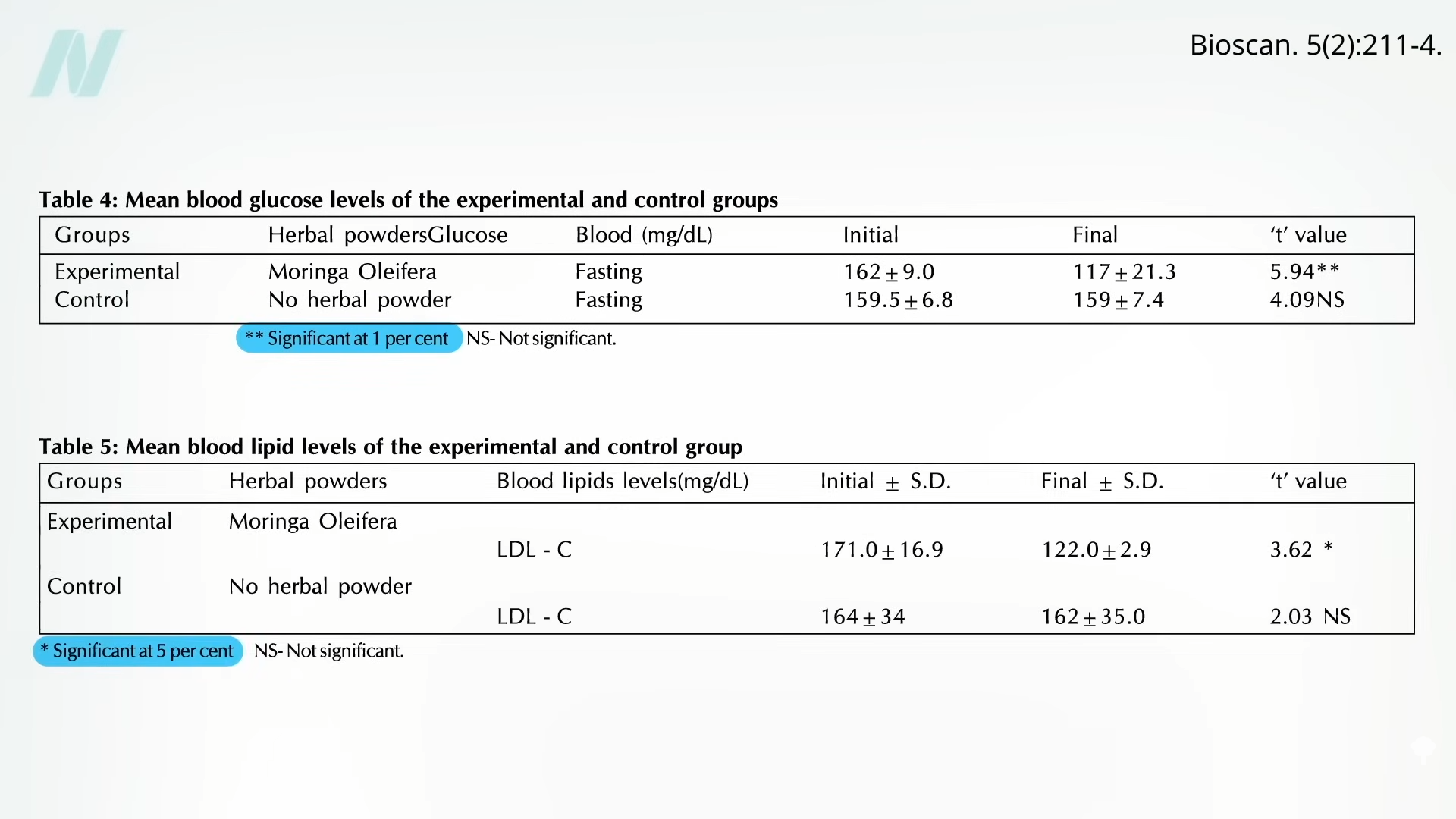
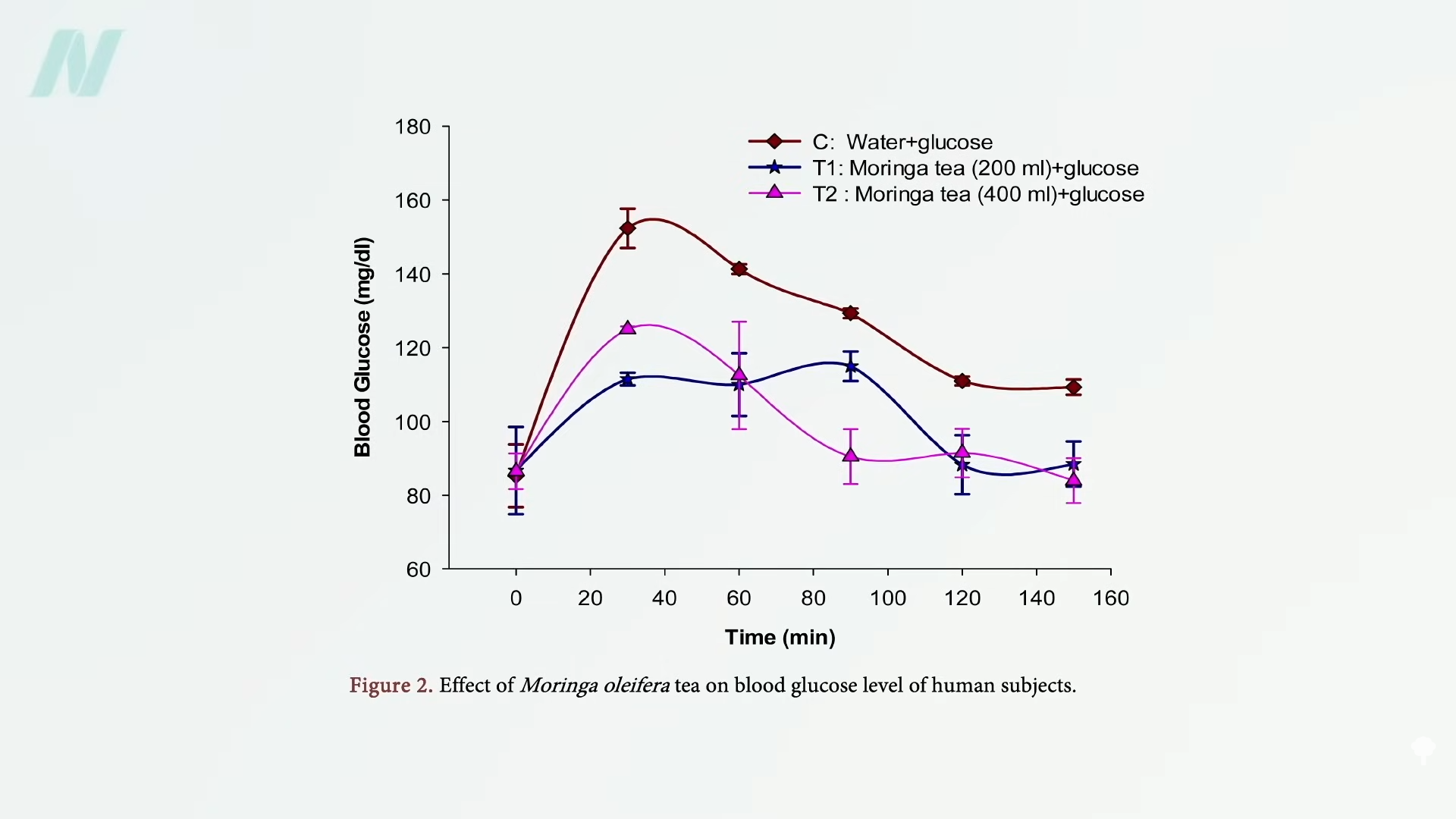 People with diabetes were
People with diabetes were