[ad_1]
What is the recommended diet for treating leaky gut? Which foods and food components can boost the integrity of our intestinal barrier?
Our intestinal tract is the largest barrier between us and the environment. More than what we touch or breathe, what we eat is our largest exposure to the outside world. Normally, our entire gastrointestinal tract is impermeable to what’s inside of it, allowing our body to pick and choose what goes in or out. But there are things that may make our gut leaky, and the chief among them is our diet.
The standard American or Western diet can cause gut dysbiosis, meaning a disruption in our gut microbiome, which can lead to intestinal inflammation and a leaky intestinal barrier. Then, tiny bits of undigested food, microbes, and toxins can slip uninvited through our gut lining into our bloodstream and trigger chronic systemic inflammation.
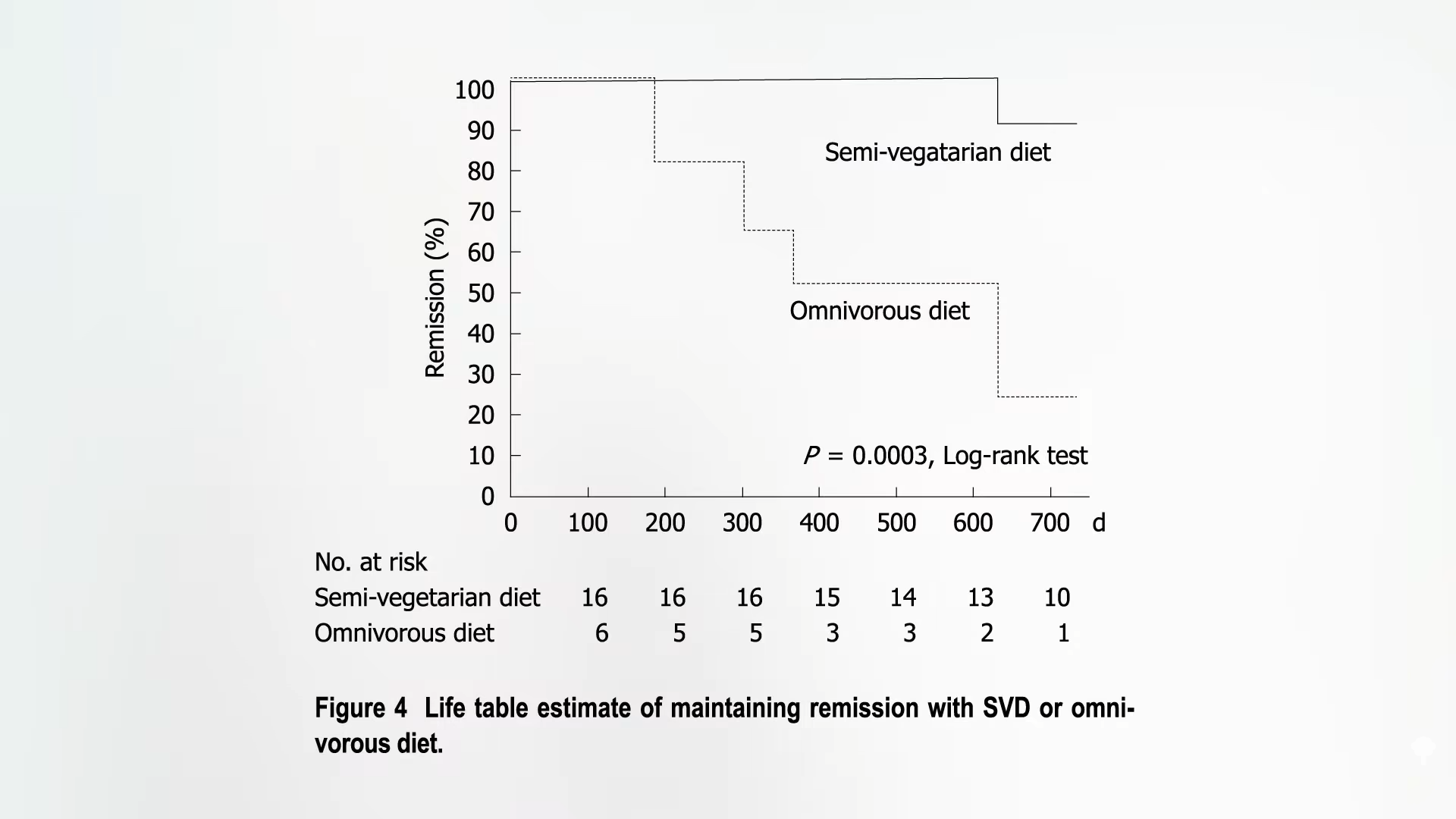
“To avoid this dysbiosis and intestinal inflammation, a predominantly vegetarian diet”—in other words, eating plants—“should be preferred.” The gut bacteria of people eating a vegetarian diet are associated with intestinal microbiome balance, high bacterial biodiversity, and integrity of the intestinal barrier. Vegetarians tend to have markedly less uremic toxins, like indole and p-cresol, and because fiber is the primary food for our gut microbiome, the gut bacteria of those eating plant-based diets have been found to produce more of the good stuff—namely short-chain fatty acids that play “a protective and nourishing role” for the cells lining our gut, “ensuring the preservation” of our intestinal barrier. Plant fiber is of “prime importance” to preserving the integrity of our intestinal barrier, but you can’t know for sure until you put it to the test.
When people with non-alcoholic fatty liver disease were given whole grains, beans, lentils, fruits, vegetables, nuts, and seeds for six months, they had a significant reduction in zonulin levels.
Zonulin is a protein responsible for the disassembly of the tight junctions between gut-lining cells and is “considered to be the only measurable biomarker that reflects an impairment of the intestinal barrier.” In other words, zonulin is a useful marker of a leaky gut. But since adding all those plants seemed to lower levels, that may “imply that appropriate fiber intake helps to maintain the proper structure and function of the intestinal barrier.” But whole healthy plant foods have a lot more than fiber. How do we know it’s the fiber? And the study didn’t even have a control group. That’s why the researchers said “gut permeability might be improved by dietary fiber” [emphasis added]. To prove cause and effect, it’d be nice to have a randomized, double-blind, crossover study where you compare the effect of the same food with or without fiber.
Such a study does, in fact, exist! A group of healthy young men was randomized to eat pasta with or without added fiber, and there was a significant drop in zonulin levels in the added-fiber group compared to both pre-intervention levels and those of the control group, as you can see below and at 2:51 in my video How to Heal a Leaky Gut with Diet.
So, fiber does indeed appear to improve gut leakiness.
Are there any plant foods in particular that may help? Curcumin, the yellow pigment in the spice turmeric, can help prevent the intestinal damage caused by ibuprofen-type drugs in rats. Similar protection was noted for the broccoli compound sulforaphane in mice. There are no human studies on broccoli yet, but there was a study on three days of the equivalent of about 2 to 3 teaspoons a day of turmeric, which did reduce markers of gastrointestinal barrier damage and inflammation caused by exercise compared to a placebo. Less turmeric may work, too, but no smaller doses have been put to the test.
If you ask alternative medicine practitioners what treatments they use for a leaky gut, number one on the list—after reducing alcohol consumption—is zinc. You can see the list below and at 3:42 in my video.
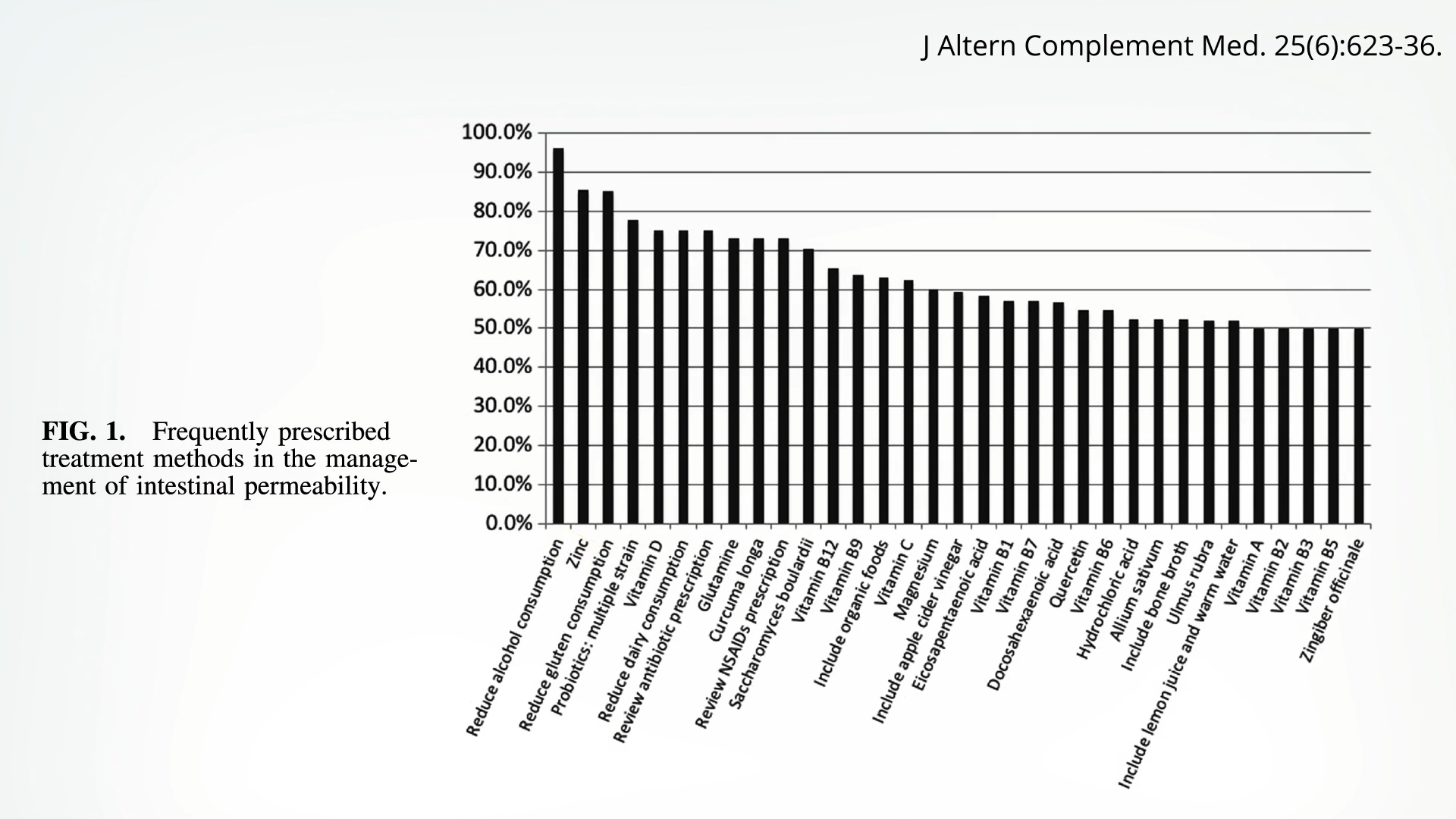
Zinc doesn’t just protect against aspirin-like drug-induced intestinal damage in rats; when put to the test in a randomized trial of healthy adults, the same thing was found. Five days of 250 mg of indomethacin, an NSAID drug, “caused a threefold rise in gut permeability,” as one would expect from that class of drugs. But this rise in permeability did not occur when participants also took zinc, “strongly suggesting a small-intestinal protective effect.” The dose they used was massive, though—75 mg a day, which is nearly twice the tolerable upper daily limit for zinc. What about getting zinc just at regular doses from food?
A significant improvement in gut leakiness was found even with a dose of just 3 mg of zinc, suggesting that even relatively low zinc supplementation may work. You can get an extra 3 mg of zinc in your daily diet by eating a cup (200g) of cooked lentils.
Doctor’s Note
For more on preventing gut dysbiosis and leaky gut, check out Flashback Friday: Gut Dysbiosis: Starving Our Microbial Self and Avoid These Foods to Prevent a Leaky Gut.
[ad_2]
Michael Greger M.D. FACLM
Source link