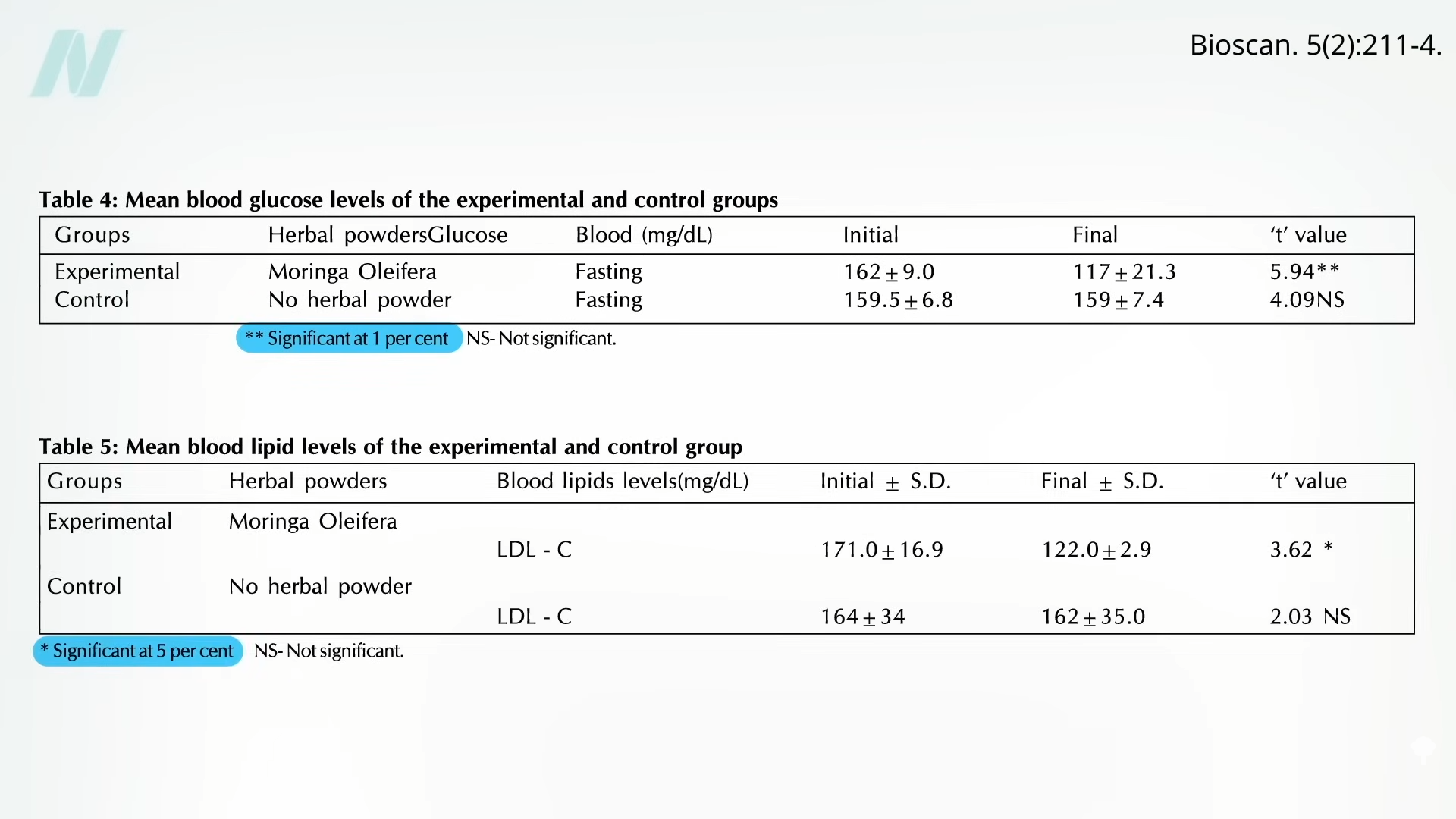
CAPE TOWN, South Africa — Twice-yearly shots used to treat AIDS were 100% effective in preventing new infections in women, according to study results published Wednesday.
There were no infections in the young women and girls that got the shots in a study of about 5,000 in South Africa and Uganda, researchers reported. In a group given daily prevention pills, roughly 2% ended up catching HIV from infected sex partners.
“To see this level of protection is stunning,” said Salim Abdool Karim of the injections. He is director of an AIDS research center in Durban, South Africa, who was not part of the research.
The shots made by U.S. drugmaker Gilead and sold as Sunlenca are approved in the U.S., Canada, Europe and elsewhere, but only as a treatment for HIV. The company said it is waiting for results of testing in men before seeking permission to use it to protect against infection.
Read More: How Depression Can Affect Dental Hygiene
The results in women were published Wednesday in the New England Journal of Medicine and discussed at an AIDS conference in Munich. Gilead paid for the study and some of the researchers are company employees. Because of the surprisingly encouraging results, the study was stopped early and all participants were offered the shots, also known as lenacapavir.
While there are other ways to prevent HIV infection, like condoms or daily pills, consistent use has been a problem in Africa. In the new study, only about 30% of participants given Gilead’s Truvada or Descovy prevention pills actually took them—and that figure dropped over time.
The prospect of a twice-a-year shot is “quite revolutionary news” for our patients, said Thandeka Nkosi, who helped run the Gilead research at the Desmond Tutu Health Foundation in Masiphumelele, South Africa. “It gives participants a choice and it just eliminates the whole stigma around taking pills” to prevent HIV.
Experts working to stop the spread of AIDS are excited about the Sunlenca shots but are concerned Gilead hasn’t yet agreed on an affordable price for those who need them the most. The company said it would pursue a “voluntary licensing program,” suggesting that only a select number of generic producers would be allowed to make them.
“Gilead has a tool that could change the trajectory of the HIV epidemic,” said Winnie Byanyima, executive director of the Geneva-based U.N. AIDS agency.
Read More: Why It Took So Long to Finally Get an RSV Vaccine
She said her organization urged Gilead to share Sunlenca’s patent with a U.N.-backed program that negotiates broad contracts allowing generic drugmakers to make cheap versions of drugs for poorer countries worldwide. As an HIV treatment, the drug costs more than $40,000 a year in the U.S., although what individuals pay varies.
Dr. Helen Bygrave of Doctors Without Borders said in a statement that the injections could “reverse the epidemic if it is made available in the countries with the highest rate of new infections.” She urged Gilead to publish a price for Sunlenca that would be affordable for all countries.
In a statement last month, Gilead said it was too early to say how much Sunlenca would cost for prevention in poorer countries. Dr. Jared Baeten, Gilead’s senior vice president of clinical development, said the company was already talking to generics manufacturers and understood how “deeply important it is that we move at speed.”
Another HIV prevention shot, Apretude, which is given every two months, is approved in some countries, including in Africa. It sells for about $180 per patient per year, which is still too pricey for most developing countries.
Byanyima said the people who need long-lasting protection the most include women and girls who are victims of domestic violence and gay men in countries where same-sex relationships are criminalized. According to UNAIDS, 46% of new HIV infections globally in 2022 were in women and girls, who were three times more likely to get HIV than males in Africa.
Byanyima compared the news about Sunlenca to the discovery decades ago of AIDS drugs that could turn HIV infection from a death sentence into a chronic illness. Back then, South African President Nelson Mandela suspended patents to allow wider access to the drugs; the price later dropped from about $10,000 per patient per year to about $50.
Read More: Dengue Is Rising in the U.S. Here’s How to Protect Yourself
Olwethu Kemele, a health worker at the Desmond Tutu Health Foundation, predicted the shots could boost the number of people coming in for HIV prevention and slow the virus’ spread. She said young women often hide the pills to avoid questions from boyfriends and family members. “It makes it hard for the girls to continue,” she said.
In a report on the state of the global epidemic released this week, UNAIDS said that fewer people were infected with HIV in 2023 than at any point since the late 1980s. Globally, HIV infects about 1.3 million people every year and kills more than 600,000, mainly in Africa. While significant progress has been made in Africa, HIV infections are rising in Eastern Europe, Latin America and the Middle East.
In other research presented at the AIDS conference, Andrew Hill of the University of Liverpool and colleagues estimated that once production of Sunlenca is expanded to treat 10 million people, the price should fall to about $40 per treatment. He said it was critical that health authorities get access to Sunlenca as soon as possible.
“This is about as close as you can get to an HIV vaccine,” he said.
___
Cheng reported from London.
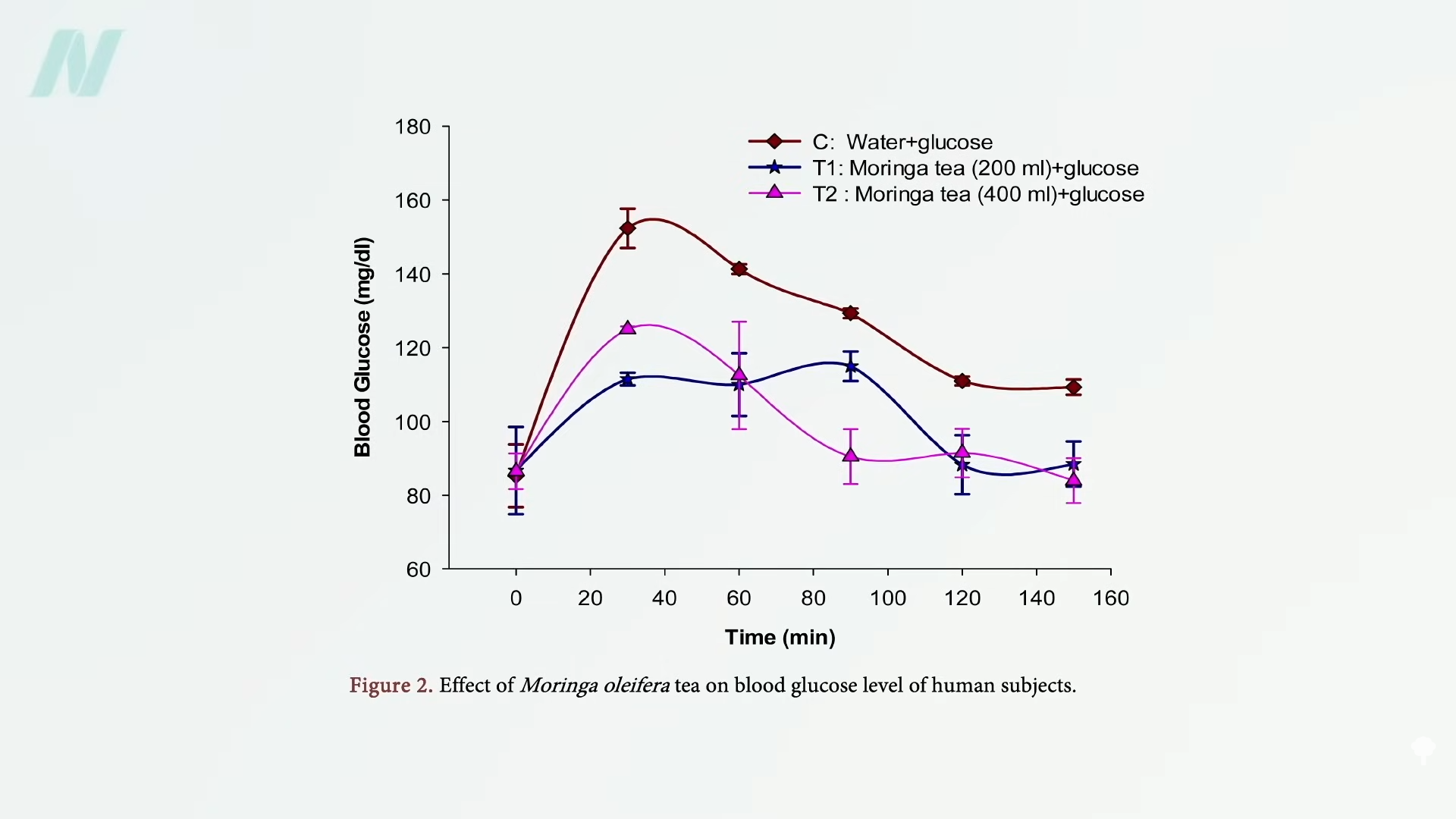 People with diabetes were
People with diabetes were