[ad_1]
The painful, inherited blood disorder afflicts mostly Black people in the U.S.
[ad_2]

[ad_1]
The painful, inherited blood disorder afflicts mostly Black people in the U.S.
[ad_2]

[ad_1]
Consumers who eat cantaloupe are advised to be on high alert following a spate of government warnings over a deadly salmonella outbreak linked to the fruit.
U.S. health officials have ordered sweeping recalls of potentially contaminated whole and pre-sliced cantaloupes over the past few weeks, in addition to urging consumers on Thursday to toss out any products containing the melon, recall notices from officials show. The flurry of warnings has prompted major grocery sellers such as Kroger, Trader Joe’s and Walmart to continue removing products containing the melon from store shelves.
Bacteria-harboring cantaloupes have been linked to at least 117 illnesses, including 61 hospitalizations and two deaths across 34 U.S. states, and those numbers could grow, the Centers for Disease Control and Prevention said Thursday in a statement.
Here’s what to know about the latest rash of cantaloupe recalls, and how to know if you should save or toss that fruit in your fridge.
Following an outbreak of severe bacterial infections linked to the fruits, the CDC is warning consumers to steer clear of pre-cut cantaloupe if they are unsure of whether it is from a distributor whose product has been recalled.
The guidance follows previous orders by U.S. health officials to recall whole cantaloupes from several brands, including Malichita and Rudy, which prompted several nationwide grocery chains to recall their own products containing pre-cut cantaloupes. Most recently, Sprouts Farmers Market and Trader Joe’s on Wednesday pulled select fresh-cut products made from whole cantaloupes off their shelves, according to an FDA notice.
Cut Fruit Express of Inver Grove Heights, Minnesota, on Wednesday recalled products containing cantaloupe, including Caribou Coffee Fruit Mix CHPG 6.5oz; Cut Fruit Express Brand 6.5oz, 15oz, 16oz, 32oz packages of fruit mix; and food service packages of 5lb-tray, 10lb-bag, 25lb-Pail, all of which could be contaminated with salmonella.
Whole fresh cantaloupes with Malichita, Rudy, “4050” and “Product of Mexico/produit du Mexique” labels are not safe to consume and should be thrown away, the FDA said.
In addition, products containing pre-cut cantaloupe such as fruit salads may pose a salmonella risk to consumers, according to the agency. Since November, there have been at least three waves of recalls issued over pre-cut cantaloupe by major grocery stores, including Kroger, Trader Joe’s, Sprouts Farmer Market, Aldi, and Walmart, according to the FDA.
The FDA is advising consumers to toss out any cantaloupe that cannot be identified by brand.
“If you cannot tell if your cantaloupe, including pre-cut cantaloupe or products containing pre-cut cantaloupe is part of the recall, do not eat or use it and throw it away,” the agency said Thursday in a statement.
In addition to throwing out the melon, that FDA advises people to wash any surfaces that may have come into contact with the cantaloupe, using hot, soapy water or a dishwasher, the agency said.
Salmonella, or salmonellosis, is a bacterial infection that affects the intestinal tract, according to the Mayo Clinic, a nonprofit medical research center. Symptoms include diarrhea, fever and stomach cramps, which usually begin between eight and 72 hours after exposure to the bacteria.
Not everyone who contracts salmonella experiences symptoms, according to the Mayo Clinic. Children under age five, the elderly and those with weakened immune systems are more at risk of developing severe illnesses that require medical care or hospitalization.
People who get salmonella should rest and drink plenty of fluids to avoid dehydration, according to the Cleveland Clinic. The infection usually resolves on its own in a few days.
[ad_2]

[ad_1]
WASHINGTON, November 29, 2023 (Newswire.com)
–
Today, the Food and Drug Administration (FDA) promoted Dr. Namandjé Bumpus, Ph.D., to Principal Deputy Commissioner. Farah K. Ahmed, President and CEO of Fragrance Creators Association (Fragrance Creators), shared the following: “On behalf of Fragrance Creators’ staff and member companies, I want to extend my warmest congratulations to Dr. Bumpus for this well-deserved promotion. As the FDA noted in its announcement, Dr. Bumpus is a champion of science, an advocate for public health, and an impressive scientist; we wholeheartedly agree.”
The announcement came as the FDA works to develop, advance, and implement key public health initiatives, including the Modernization of Cosmetics Regulation Act (MoCRA). “Fragrance Creators values collaborating with Dr. Bumpus as we continue to serve as a resource to the Agency. She has been a thoughtful leader and a scientist of the highest caliber,” said Ahmed. “Personally, as a former FDA employee, I am delighted to see the agency also prioritize diversity — an inspiration for all women, especially women of color — in this role.”
Fragrance plays an important role in home care, cosmetics, and personal care products, products that impact everyday life, nurture skin health and hygiene, empower self-expression and confidence, and deliver generational delight to billions of people every day. Fragrance Creators recognizes the FDA’s and Dr. Bumpus’ commitment to advancing public health predicated on best-in-class science, as our members remain dedicated to ensuring we continue to support the agency — for people, perfume, and the planet.
Fragrance Creators looks forward to continuing our engagement with Dr. Bumpus as she transitions from Chief Scientist into her new role as Principal Deputy Commissioner, succeeding Dr. Janet Woodcock, M.D., when Dr. Woodcock retires early next year. “We also want to thank Dr. Woodcock for the tremendous advancements she has made for public health, science, and women in science,” said Ahmed. “We wish her well in this next chapter.”
###
Fragrance Creators Association is the trade association representing the majority of fragrance manufacturing in North America. We also represent fragrance-related interests along the value chain. Fragrance Creators’ member companies are diverse, including large, medium, and small-sized companies that create, manufacture, and use fragrances and scents for home care, personal care, home design, fine fragrance, and industrial and institutional products, as well as those that supply fragrance ingredients, including natural extracts and other raw materials that are used in perfumery and fragrance mixtures. Fragrance Creators established and administers the Congressional Fragrance Caucus, ensuring ongoing dialogue with members of Congress and staff. Fragrance Creators also produces The Fragrance Conservatory, the comprehensive digital resource for high-quality information about fragrance — www.fragranceconservatory.com. Learn more about Fragrance Creators at fragrancecreators.org — for people, perfume, and the planet.
Source: Fragrance Creators Association
[ad_2]

[ad_1]
The FDA has expanded its list of eye drops recalled in 2023 because the products could be tainted with bacteria.
Store-brand products sold at retailers including CVS, Rite Aid, Target and Walmart are included in the latest recall, posted on Wednesday by the federal safety agency. The recall by Kilitch Healthcare India cited “safety concerns” after FDA investigators found unsanitary conditions at the facility where the products were made.
The FDA has cited a “risk of eye infections that could result in partial vision loss or blindness” in cautioning consumers against using the now-recalled products in late October. FDA investigators found bacteria in samples taken from the production plant, the agency noted in its warning. As of Nov. 13, Kilitch said it hasn’t received any reports of adverse health effects linked to the recall.
Many of the eye drops are likely no longer in stores, as the FDA said CVS, Rite Aid and Target were removing the products from store shelves and websites.
The recall notice urged distributors and retailers to stop selling the products and for consumers to return them.
Amazon said its website was no longer offering seven eye drop products after the FDA warned the e-commerce giant that their distribution violated federal regulations and raised public health concerns.
The pulled products named in the agency’s letter are: Similasan Pink Eye Relief, The Goodbye Company Pink Eye, Can-C Eye Drops, Optique 1 Eye Drops, OcluMed Eye Drops, TRP Natural Eyes Floaters Relief and Manzanilla Sophia Chamomile Herbal Eye Drops.
“We require all products offered in our store to comply with applicable laws and regulations. The products in question have been investigated and removed,” an Amazon spokesperson told CBS MoneyWatch on Friday in an emailed statement.
The FDA’s warning prompted Cardinal Health in early November to recall six Leader brand eye products, while Harvard Drug Group recalled Rugby Laboratories brand eye drops.
Those warnings included the agency in August advising people not to buy “Dr. Berne’s MSM Drops 5% Solution” and “LightEyez MSM Eye Drops – Eye Repair,” saying they presented serious health risks, including vision- and life-threatening infections.
The recalls are among eight such actions involving the FDA and eye drops this year, starting with news in early February that the Centers for Disease Control and Prevention (CDC) was investigating a cluster of infections related to artificial tears.
Federal inspectors found dozens of problems at Indian manufacturer Global Pharma Healthcare amid the multistate outbreak of rare and extensively drug-resistant bacteria linked by eye drops made by the company, which were branded as EzriCare and Delsam Pharma.
The CDC ultimately identified 81 patients in 18 states. Of that that number, four people died, 14 reported vision loss and another four had to have their eyeballs surgically removed, the agency said in its last update in May.
Eye drops must be sterile to be safe, and therefore need “much finer oversight on the manufacturing side” to ensure the products aren’t contaminated, Carri Chan, a business professor at Columbia University, recently told CBS MoneyWatch.
The FDA maintains an updated list of eye drop products that people should get rid of. Users can also consult an eye drop manufacturer’s website to check if a product has been recalled.
Dr. Christopher Starr, a spokesperson for the American Academy of Ophthalmology, said people should seek medical help immediately if they have adverse reactions to eye drops, such as discharge, redness or pain, which can indicate an infection. People should also check a product’s expiration date before use.
Consumers with questions about the recall can contact Kilitch at regulatory@kilitchhealthcare.com or Velocity Pharma, Kilitch’s distributor, at regulatory@velocitypharma.com. Eye drop users who experience adverse reactions can file a report with the FDA online or by downloading and mailing a pre-addressed form.
—Elizabeth Napolitano contributed to this report.
[ad_2]

[ad_1]
Another eye drop recall is pulling 27 products from store shelves, including store-brand products sold at retailers such as CVS, Rite Aid, Target and Walmart, with the recall coming about three weeks after the U.S. Food and Drug Administration warned people not to buy or use the eye drops.
Kilitch Healthcare India is recalling eye drops with expiration dates ranging from November 2023 to September 2025, citing “safety concerns” due to FDA investigators finding unsanitary conditions at the facility where the products were made, according to a news release posted Wednesday by the agency. You can see the full list of recalled eye drops here.
The FDA cited a “risk of eye infections that could result in partial vision loss or blindness” in urging against use of the products in late October, with the agency saying its investigators found unsanitary conditions and “positive bacterial test results” from areas of an unidentified manufacturing facility. As of Nov. 13, Kilitch said it hasn’t received any reports of adverse health effects linked to the recall.
Some of the eye drops may have already been removed from stores, as earlier this month the FDA said CVS, Rite Aid and Target were taking the products off their shelves and websites.
The recall notice said that distributors and retailers that have any of the recalled products should stop distributing them. Consumers, meanwhile, should stop using the recalled eye drops and return the products to where they purchased them, the FDA said.
Bloomberg News reported that Kilitch produced the eye drops in an unsanitary factory in India where some workers went barefoot and others made up test results that purported to show the products were sterile.
The FDA’s warning prompted Cardinal Health in early November to recall six Leader brand eye products, while Harvard Drug Group recalled Rugby Laboratories brand eye drops.
The FDA earlier this year announced a spate of recalls of eye drop products linked to four deaths and multiple cases of vision loss. In August, it warned against using two additional eye drop products because of the risk of bacterial or fungal contamination.
Eye drop products, which must be sterile in order to be safe, require “much finer oversight on the manufacturing side in order to ensure that there’s no contamination whatsoever,” Carri Chan, a business professor at Columbia University, told CBS MoneyWatch last week in reference to the spate of recalls.
The FDA keeps an updated list of eye drop products that consumers should discard. Users can also consult an eye drop manufacturer’s website to check if a product has been recalled.
Dr. Christopher Starr, a spokesman for the American Academy of Ophthalmology, said people should seek medical help immediately if they experience adverse reactions to their eye drops, told CBS MoneyWatch. Signs of an infection typically include discharge, redness or pain. Consumers should also check a product’s expiration date before use.
Consumers with questions about the recall can contact Kilitch at regulatory@kilitchhealthcare.com or Velocity Pharma, Kilitch’s distributor, at regulatory@velocitypharma.com. Eye drop users who experience adverse reactions can file a report with the FDA online or by downloading and mailing a pre-addressed form.
—Elizabeth Napolitano contributed to this report.
[ad_2]

[ad_1]
Studies show that many doctors either tend to overestimate the amount of weight that can be lost with obesity drugs or are simply clueless.
Current options for weight-loss medications include the ridiculously named Qsymia, a combination of phentermine (the phen in fen-phen) and topiramate, a drug that can cause seizures if you stop it abruptly. Qsymia was “explicitly rejected” multiple times for safety reasons in Europe “because of concerns about the medicine’s long-term effects on the heart and blood vessels” but, at the time of making my video Are Weight Loss Pills Effective?, remains available for sale in the United States. Belviq is in a similar boat—allowed in the United States but not in Europe due to “concerns about possible cancers, psychiatric disorders, and heart valve problems…”
Belviq is sold in the United States for about $200 a month. If you think that’s a lot, there’s Saxenda, which requires daily injections and is listed at the low, low price of only $1,281.96 for a 30-day supply. It carries a black box warning, the FDA’s strictest caution about potentially life-threatening hazards, for thyroid cancer risk. Paid consultants and employees of the company that makes it argue the greater number of breast tumors found among drug recipients may be due to “enhanced ascertainment,” meaning easier breast cancer detection just due to the drug’s effectiveness.
Contrave is another option if you ignore its black box warning about a potential increase in suicidal thoughts. Then there’s Alli, the drug that causes fat malabsorption, thereby resulting in side effects “including fecal urgency, oily stool, flatus with discharge, and fecal incontinence”—Alli can be your ally in anal leakage. The drug evidently “forces the patient to use diapers and to know the location of all the bathrooms in the neighborhood in an attempt to limit the consequences of urgent leakage of oily fecal matter.” A Freedom of Information Act exposé found that although company-sponsored studies claimed that “all adverse events were recorded,” one trial apparently conveniently failed to mention 1,318 of them.
What’s a little bowel leakage, though, compared to the ravages of obesity? As with anything in life, it’s all about risks versus benefits. However, in an analysis of more than a hundred clinical trials of anti-obesity medications that lasted up to 47 weeks, drug-induced weight loss never exceeded more than nine pounds. That’s a lot of money and a lot of risk for just a few pounds. Since you aren’t treating the underlying cause—a fattening diet—when people stop taking these drugs, the weight tends to come right back, so you’d have to take them every day for the rest of your life. But people do stop taking them. Using pharmacy data from a million people, most Alli users stopped after the very first purchase and most Meridia users didn’t even make it three months. Taking weight-loss meds is so disagreeable that 98 percent of users stopped taking them within the first year.
Studies show that many doctors tend to overestimate the amount of weight that can be lost with these drugs or are simply clueless. One reason may be that some clinical practice guidelines go out of their way to advocate prescribing medications for obesity. Are they seriously recommending drugging a third of Americans—more than 100 million people? You may not be surprised to learn that the principal author of the guidelines has a “significant financial interest or leadership position” in six separate pharmaceutical companies that all (coincidently) work on obesity drugs. In contrast, independent expert panels, like the Canadian Task Force on Preventive Health Care, explicitly recommend against weight-loss drugs, given their poor track record of safety and efficacy.
In case you missed my related video, check out Are Weight Loss Pills Safe?.
As with all lifestyle diseases, it’s better to treat the underlying cause, which, in the case of obesity, is a fattening diet. For an example of what’s possible with a healthy diet intervention, see Flashback Friday: The Weight Loss Program That Got Better with Time.
Check out the related videos below for more about weight loss.
[ad_2]
Michael Greger M.D. FACLM
Source link

[ad_1]
Twenty-two children in 14 states have reportedly fallen ill after eating applesauce pouches “tainted” with lead. According to AP News, the CDC issued a health alert for parents and doctors on Monday, November 13.
According to the CDC, multiple states have reported cases to the US Food and Drug Administration (FDA) linking “high blood lead levels” to children who have eaten “recalled cinnamon-containing applesauce products.”
The FDA reports that the affected applesauce products include “Recalled WanaBana apple cinnamon fruit puree pouches, Recalled Schnucks-brand cinnamon-flavored applesauce pouches and variety pack and Recalled Weis-brand cinnamon applesauce pouches.”
🚨Additional Recall Alert for Applesauce Puree Pouches Due to Lead Contamination
Recall now includes WanaBana apple cinnamon fruit puree pouches, Schnucks brand cinnamon-flavored applesauce pouches & variety pack, & Weis brand cinnamon applesauce pouches.https://t.co/8NksSwKSAu pic.twitter.com/CesMd9MJGo
— Tompkins County Whole Health (@TCWholeHealth) November 8, 2023
According to AP News, 22 children have been sickened by the lead-tainted applesauce pouches thus far. The outlet reports that the children are between the ages of between the ages of 1-3.
The FDA reports that children who have been impacted by lead poisoning may not have “obvious immediate symptoms.” However, short-term exposure to lead can lead to symptoms such as headaches, vomiting, abdominal pain/colic, and anemia in children.
Long-term exposure can lead to muscle aches, burning or weakness, constipation, tremors, and even weight loss, among other symptoms.
The CDC adds that the effects of lead poisoning in children “may continue into adulthood.”
According to the CDC, the 22 reported cases come from 14 states.
“Alabama, Arkansas, Louisiana, Maryland, Missouri, New Mexico, New York, North Carolina, Ohio, Pennsylvania, South Carolina, Tennessee, Texas, and Washington,” the CDC‘s official website reads as the total 14.
Furthermore, AP News notes that the impacted applesauce products have been sold in-store and online. The FDA states that consumers should not “eat, sell, or serve” the WanaBana, Schnucks, or Weis applesauce pouches but instead immediately “discard them.”
The FDA also recommends that consumers thoroughly search their homes for these products as they may have been stored away due to their longer shelf life.
Furthermore, the FDA recommends that parents who are suspicious their children may have eaten the impacted products bring them to a medical professional for blood testing. The association also urges parents to immediately call a doctor if their children show signs of “lead toxicity.”
The association adds, “updates to this advisory will be provided as they become available.”
[ad_2]
Jadriena Solomon
Source link

[ad_1]
Why don’t more people take the weight-loss medications currently on the market?
Despite the myriad menus of FDA-approved medications for weight loss, they’ve only been prescribed for about 1 in 50 patients with obesity. We tend to worship medical magic bullets in the United States, so what gives? As I discuss in my video Friday Favorites: Are Weight-Loss Supplements Safe and Effective?, one of the reasons anti-obesity drugs are so “highly stigmatized” is that, historically, they’ve been anything but magical and the bullets have been blanks—or worse.
To date, most weight-loss drugs that were initially approved as safe have since been pulled from the market for unforeseen side effects that turned them into a “threat to public health.” As you may remember from my video Brown Fat: Losing Weight Through Thermogenesis, it all started with DNP, a pesticide with a promise to safely melt away fat that melted away people’s eyesight instead. (That actually helped lead to the passage of the landmark Food, Drug, and Cosmetic Act in 1938.) Thanks to the internet, DNP has made a comeback with “predictably lethal results.”
Then came the amphetamines. Currently, more than half a million Americans may be addicted to amphetamines like crystal meth, but the “original amphetamine epidemic was generated by the pharmaceutical industry and medical profession.” By the 1960s, drug companies were churning out about 80,000 kilos of amphetamines a year, which is nearly enough for a weekly dose for every man, woman, and child in the United States. Billions of doses a year were prescribed for weight loss, and weight-loss clinics were raking in huge profits. A dispensing diet doctor could buy 100,000 amphetamine tablets for less than $100, then turn around and sell them to patients for $12,000.
At a 1970 Senate Hearing, Senator Thomas Dodd (father of “Dodd-Frank” Senator Chris Dodd) suggested that America’s speed freak problem “was no by means an ‘accidental development’: ‘Multihundred million dollar advertising budgets, frequently the most costly ingredient in the price of a pill, have, pill by pill, led, coaxed and seduced post-World War II generations into the ‘freaked-out’ drug culture…’” I’ll leave drawing the Big Pharma parallels to the current opioid crisis as an exercise for the viewer.
Aminorex was a widely-prescribed appetite suppressant before it was pulled for causing lung damage. Eighteen million Americans were on fen-phen before it was pulled from the market for causing severe damage to heart valves. Meridia was pulled for heart attacks and strokes, Acomplia was pulled for psychiatric side effects, including suicide, and the list goes on, as you can see below and at 2:51 in my video.
The fen-phen debacle resulted in “some of the largest litigation pay-outs ever seen in the pharmaceutical industry, with individual amounts of up to US$200,000 and a total value of ~US$14 billion,” but that’s all baked into the formula. If you read the journal PharmacoEconomics (and who doesn’t!), you may be aware that a new weight-loss drug may injure and kill so many that “expected litigation cost” could exceed $80 million, but Big Pharma consultants estimate that if it’s successful, the drug could bring in more than $100 million, so do the math.
What does work for weight loss? I dive deep into that and more in How Not to Diet. For more of my videos on weight loss, check out the related videos below.
[ad_2]
Michael Greger M.D. FACLM
Source link

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
Remember the “epidemic” of underage nicotine vaping? For years, activists, politicians, and public health officials have been warning that a surge in e-cigarette use by teenagers would hook a generation of young people on nicotine and encourage them to smoke.
That never happened, as new federal survey data confirm. But policies adopted in response to that overblown threat continue to undermine the harm-reducing potential of vaping products by making them less attractive to current and former smokers.
According to the latest National Youth Tobacco Survey, which is overseen by the Centers for Disease Control and Prevention (CDC), 10 percent of high school students reported past-month e-cigarette use in 2023, down from 14 percent last year and more than 27 percent in 2019. Among middle school students, the 2023 rate was 4.6 percent, less than half the 2019 rate.
How many of those past-month vapers might reasonably be described as addicted to nicotine? A quarter of them—less than 2 percent of all respondents—reported vaping every day in the previous month, meaning that, as usual, the vast majority were occasional users.
This does not look like an epidemic of nicotine addiction. Nor did the fear that vaping would lead to smoking pan out.
Even at the peak of underage vaping, the downward trend in adolescent smoking not only continued but accelerated. In the 2023 survey, less than 2 percent of high school students reported smoking cigarettes in the previous month—down from 16 percent in 2011 and (according to a survey of 10th- and 12th-graders) more than 30 percent in 1997.
The CDC describes vaping as “tobacco product use,” even though e-cigarettes do not contain tobacco, and lumps it in with smoking, even though it does not involve combustion. But while the CDC is loath to admit it, the shift from smoking to vaping—in any age group—is indisputably an improvement in terms of health risks.
The Food and Drug Administration (FDA) acknowledges that vaping is far less hazardous than smoking, and it supposedly is committed to maintaining the availability of what it calls “electronic nicotine delivery systems” (ENDS) as a potentially lifesaving alternative for cigarette smokers. Yet to deter underage use, the FDA so far has approved only tobacco-flavored ENDS, even though former smokers overwhelmingly prefer other flavors.
That policy makes ENDS less appealing to people who already have switched from smoking to vaping or might be interested in doing so. The results are predictable: A recent FDA-supported study of sales data from across the country found that state and local restrictions on ENDS flavors were associated with increased purchases of conventional cigarettes.
“We find that ENDS flavor policies reduce flavored ENDS sales as intended, but also increase cigarette sales across age groups,” the researchers reported. “As cigarettes are much more lethal than ENDS, the high rate of substitution estimated here suggests that, on net, any population health benefits of ENDS flavor policies are likely small or even negative.”
Although “flavored ENDS products remain widely available in states that do not prohibit their sales,” the study’s authors noted, the FDA seems to be “paving a path towards a de facto national ENDS flavor prohibition.” That policy, they said, entails an “inequitable tradeoff” because it “prioritizes youth over the 11.2% of US adults [who] smoke.”
Since the FDA has not made a serious effort to enforce its de facto ban against the thousands of suppliers who are theoretically violating it, adults can still purchase ENDS in a wide variety of flavors from vape shops, tobacconists, and online vendors. Preserving those options for adults is consistent with efforts to reduce underage consumption, as the ongoing decline in adolescent vaping shows.
The flavor restrictions embraced by regulators and legislators threaten to harm public health in the name of protecting it. To save teenagers from an exaggerated danger, bureaucrats and politicians are sacrificing the interests, and perhaps the lives, of adult smokers across the country.
© Copyright 2023 by Creators Syndicate Inc.
[ad_2]
Jacob Sullum
Source link

[ad_1]
Last month, Gov. Gavin Newsom signed into law California’s Food Safety Act banning four ingredients that are linked to health risks. These substances — red dye no. 3, propyl paraben, brominated vegetable oil and potassium bromate, currently found in some candies, sodas and baked goods — will not be allowed in the state’s foods starting in 2027. All four are banned from foods in the European Union (which only allows red no. 3 in candied and cocktail cherries), but, California aside, they remain perfectly legal in the U.S.
New York is considering a similar law that would also ban a fifth substance previously included in California’s law — titanium dioxide, which is used in Skittles. That’s why the California measure got dubbed the “Skittles ban” (a name that stuck even after titanium dioxide was cut from the draft).
California is the first state to go beyond Food and Drug Administration regulations by banning the other four additives. Should it have deferred to the FDA?
The challenges facing the FDA make the case for state action. Sluggish and irregular safety reviews, a fast-track ingredient approval loophole that is abused by manufacturers, and a focus on acute food poisoning over long-term diet all hinder the agency’s ability to address the growing risks associated with our food supply.
The FDA is required to review the safety of any new food additive and grant approval before it can be used. If evidence indicates that an additive is unsafe, the FDA is supposed to decline or limit its use. Three of the substances in California’s law were approved by this standard review: potassium bromate, Red Dye No. 3 and brominated vegetable oil. But the FDA is reevaluating the safety of the latter two and has proposed, though not finalized, a rule to ban brominated vegetable oil from the food supply.
The fourth substance set to be banned in California, propyl paraben, was approved through what’s effectively a loophole in the FDA system. Ingredients classified as “generally recognized as safe” (GRAS) are exempt from the additive category and thus from careful FDA review. Congress crafted this exemption to be used infrequently, primarily to keep common ingredients like salt and spices on the market without an onerous approval process. But as food companies sought to avoid the rigorous food additive review, GRAS applications piled up.
Without the resources to research the applications, and lacking further support from Congress, the FDA allowed manufacturers to skip the application and determine GRAS status with only a “voluntary notification process.” This means companies can choose whether to let the FDA know they believe their substance is GRAS — in which case FDA can affirm that decision — or they can self-affirm GRAS status and market the substance without ever notifying the FDA. Thousands of substances have entered the food supply this way. Even when companies voluntarily notify, as was the case for propyl paraben in 1984, the FDA does not conduct a full safety review to affirm GRAS status.
Since GRAS notification is voluntary, the FDA does not know all the substances in our food supply. One study found that of the 4,284 GRAS determinations made as of January 2011, just 582 were cleared through the FDA’s voluntary notification process.
Although the FDA has the authority to revoke GRAS status or an additive approval, the agency reviews the safety of greenlighted ingredients sporadically, rather than regularly — and often slowly.
Take for example, trans fat from partially hydrogenated oils, a GRAS substance used for decades in commercial baked goods and other products. A 2004 citizen petition asked the FDA to look into the safety of these oils, but it wasn’t until 2015 that the FDA determined that they were not GRAS and banned them in food starting in 2020. By the time the FDA got around to this, New York City had already banned them in restaurants (in 2006), as had California (in 2008).
The under-regulation of food additives is part of a larger challenge. FDA vetting focuses more on acute risks, such as food-borne illness, than on longer-term risks from diet. Of the agency’s more than $1 billion budget for its foods program, only 7% goes to nutrition and labeling, its major strategies to address diet-related disease. Yet while foodborne illness causes about 3,000 deaths per year, 1.5 million deaths in 2018 — more than half of all deaths that year — resulted from conditions linked to diet.
But states moving to ban substances isn’t a perfect solution either. They generally don’t have the resources to conduct comprehensive safety reviews, and it would be more efficient to beef up the FDA’s infrastructure than to duplicate costly systems across states and potentially create a confusing patchwork of bans.
We desperately need change at the federal level. The Government Accountability Office reported on flaws in the GRAS system in 2010, and the FDA has not addressed the majority of the recommendations, such as regularly reviewing the safety of GRAS substances and requiring companies to provide basic information about these substances. The FDA urgently needs additional Congressional funding to take action on food safety for all ingredients, with a particular eye toward diet-related chronic disease.
In the meantime, states like California will have to keep taking the lead on evaluating harmful ingredients and show the federal government how it can be done.
Emily Broad Leib is a clinical professor of law at Harvard Law School and faculty director of the school’s Food Law and Policy Clinic.
[ad_2]
Emily Broad Leib
Source link

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
Federal regulators are warning consumers to stop using a number of over-the-counter eye drops that may pose an infection risk, marking the latest of several alerts this year against artificial tears.
The notice applies to 26 eye drop products marketed under several store brands, such as CVS Health, Rite Aid and Target Up & Up, in addition to the Leader, Rugby and Velocity Pharma brands, the Food and Drug Administration said Friday. The eye drops may contain bacteria that could cause eye infections, potentially blinding them, the agency said.
The FDA said it has not received any reports of infections from consumers who have used the suspect products. However, the agency is recommending that manufacturers pull all potentially contaminated products from store shelves.
According to the FDA, federal investigators found harmful bacteria and “insanitary conditions” at a manufacturing facility where several eye drop products are produced.
The agency urged consumers who have purchased the potentially contaminated eye drops to discard them. The FDA said that consumers who think they have suffered an adverse reaction to the lubricants should report their concerns to the agency’s MedWatch Adverse Event Reporting program.
The alert follows a series of eye drop recalls the FDA issued earlier this year after federal health investigators found evidence that several products were contaminated with a dangerous drug-resistant bacteria known as Pseudomonas aeruginosa. Eye drops tainted with the bacteria are linked to the death of at least four people, with dozens more affected across 18 states, Centers for Disease Control and Prevention data shows.
The FDA’s latest warning applies to the following eye drop products sold throughout the U.S.
|
Retailer/ Label |
Product |
Product Information |
|
CVS Health |
Lubricant Eye Drops 15 ml (single pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
Lubricant Eye Drops 15 ml (twin pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
|
Lubricant Gel Drops 15 ml (single pack) |
Carboxymethylcellulose Sodium Eye Drops 1% w/v |
|
|
Lubricant Gel Drops 15 ml (twin pack) |
Carboxymethylcellulose Sodium Eye Drops 1% w/v |
|
|
Multi-Action Relief Drops 15 ml |
Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
|
|
Lubricating Gel drops 10 ml |
Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
|
|
Lubricant Eye Drops 10 ml (single pack) |
Propylene Glycol Eye Drops 0.6% w/v |
|
|
Lubricant Eye Drops 10 ml (twin pack) |
Propylene Glycol Eye Drops 0.6% w/v |
|
|
Mild Moderate Lubricating Eye Drops 15 ml (single pack) |
Polyethylene Glycol 400 Eye Drop ‘0.25% w/v |
|
|
Rugby (Cardinal Health) |
Lubricating Tears Eye Drops 15 ml |
Hypromellose 2910-0.3% w/v & Dextran 70- 0.1% Eye Drops |
|
Polyvinyl Alcohol 1.4% Lubricating Eye Drops 15 ml |
Polyvinyl Alcohol Eye Drops 1.4% w/v |
|
|
Leader (Cardinal Health) |
Dry Eye Relief 10 ml |
Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
|
Lubricant Eye Drops 15 ml (single pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
|
Lubricant Eye Drops 15 ml (twin pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
|
Dry Eye Relief 15 ml |
Carboxymethylcellulose Sodium Eye Drops 1% w/v |
|
|
Eye Irritation Relief 15 ml |
Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
|
|
Rite Aid |
Lubricant Eye Drops 15 ml (twin pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
Lubricant Eye Drops 10 ml (twin pack) |
Propylene Glycol Eye Drops 0.6% w/v |
|
|
Gentle Lubricant Gel Eye Drops 15 ml |
Hypromellose 0.3%, Glycerin 0.2%, Dextran 70 0.1% Eye Drops |
|
|
Lubricant Gel Drops 15 ml |
Carboxymethylcellulose Sodium Eye Drops 1% w/v |
|
|
Lubricating Gel Drops 10 ml |
Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
|
|
Multi-Action Relief Drops 15 ml |
Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
|
|
Target |
Up&Up Dry Eye Relief Lubricant Eye Drops 30 ml |
Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
|
Up&Up Extreme Relief Dry Eye 15 ml (single pack) |
Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
|
|
Up&Up Extreme Relief Dry Eye 30 ml (twin pack) |
Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
|
|
Velocity Pharma LLC |
Lubricant Eye Drop 10 ml (triple pack) |
Propylene Glycol Eye Drops 0.6% w/v |
Thanks for reading CBS NEWS.
Create your free account or log in
for more features.
[ad_2]

[ad_1]
The Food and Drug Administration (FDA) approves not only which drugs can go to market but also whether they require a prescription from a doctor. The agency gets this power from the Durham-Humphrey Amendment of 1951. Meant to protect patients, the law has turned into a license to make life difficult. Here are five low-risk drugs that the FDA could make available over the counter today.
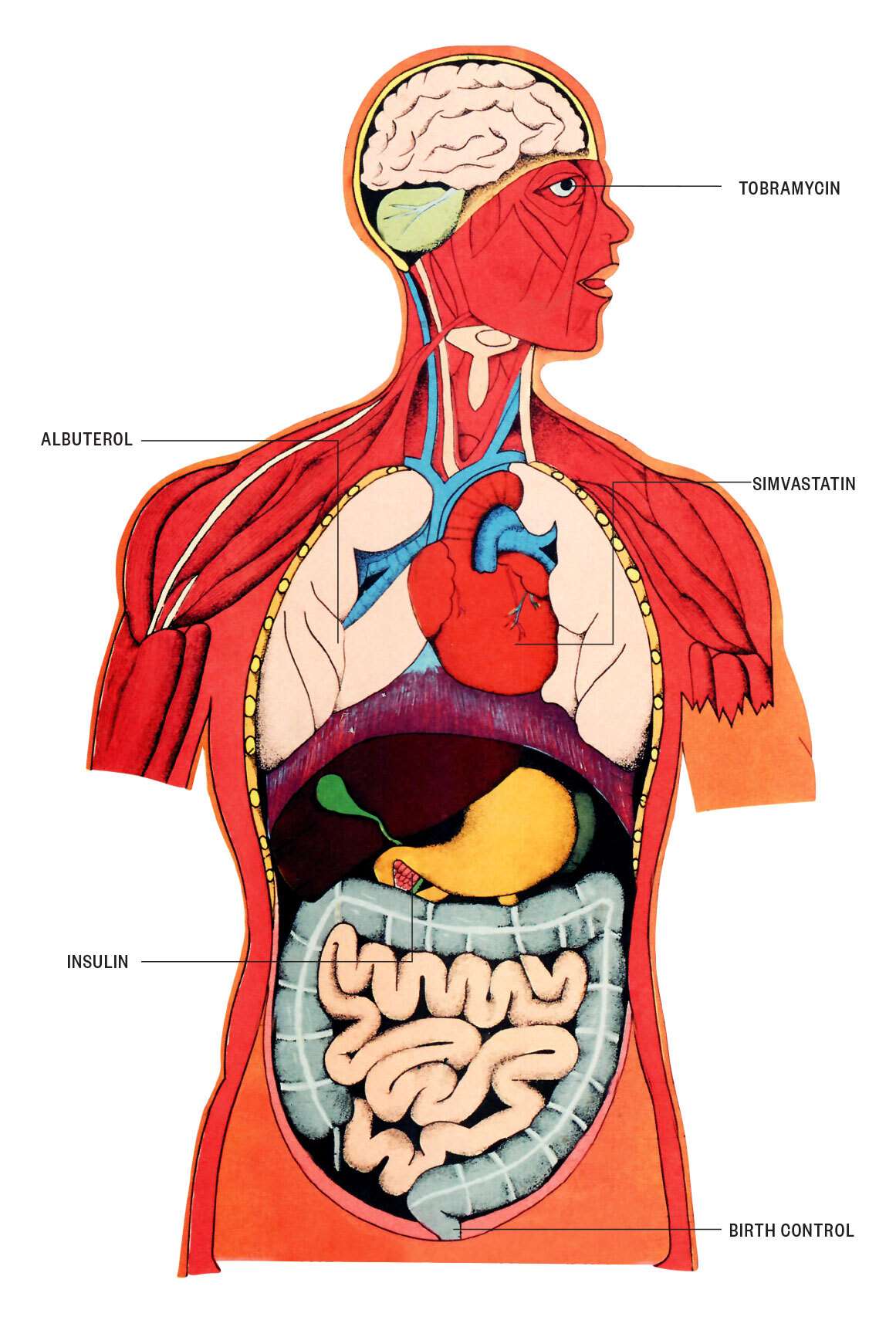
Albuterol: Research shows that albuterol is vastly superior at relieving asthma symptoms compared to other inhalers, which is why it’s offered over the counter in Singapore, Australia, Slovenia, and South Africa. It’s also available in the United Kingdom under the name Salbutamol. There is no logical safety-related reason why albuterol sits behind the counter while its competitors, the epinephrine drugs Primatene Mist and Asthmanefrin, are available without a prescription.
Insulin: Two outdated versions of insulin are sold over the counter because they were grandfathered in after the passage of the Durham-Humphrey Amendment, but those aren’t the products that today’s diabetics want or need. As insulin prices have skyrocketed in the U.S., a black market for insulin has emerged. Products like insulin can and should be sold through online retailers like Amazon, especially given the long wait times to see the medical specialists who prescribe the drug.
Tobramycin: Tobramycin eye drops are used to treat conjunctivitis, more commonly known as “pinkeye.” It’s pretty obvious when someone has pinkeye, but patients still have to go to the doctor to get a prescription for these unharmful eye drops. That often means parents either have to go to the emergency room or miss work to take their kid to the pediatrician.
Simvastatin: Heart disease is the leading cause of death in the U.S., and statins—cholesterol-lowering drugs—can reduce the risk of a heart attack and other common deaths from heart disease. Simvastatin (and other statins like lovastatin) are prescribed for cardiovascular disease. This particular drug has been over-the-counter in the U.K. since 2004 but has failed to get approval for unrestricted usage in the United States. A 2016 study published in the American Journal of Managed Care estimated that over-the-counter statin access could prevent over 250,000 major coronary events over 10 years.
Birth control: With over 9 million users, daily-use oral contraception, or the birth control pill, is the most common form of contraception in the United States. The American College of Obstetricians and Gynecologists, which represents birth control prescribers, says it “supports over-the-counter access to hormonal contraception without age restrictions.” On July 13, the FDA did approve the progestin-only Opill brand for over-the-counter use, which is a step in the right direction.
[ad_2]
Natalie Dowzicky
Source link

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
Watch CBS News
Be the first to know
Get browser notifications for breaking news, live events, and exclusive reporting.
[ad_2]

[ad_1]
The American was infamously stripped of his 2006 Tour de France win after testing positive for performance-enhancing drugs. Disgrace, downfall and eventually redemption all ensued, and he went on to launch Floyd’s of Leadville in 2016, which sells CBD products such as gummies, coffee, drink mixes and tonics.
CBD is oil or powder derived from the cannabis plant that can be added as an ingredient in food and beverages. It doesn’t contain tetrahydrocannabinol (THC), the psychoactive ingredient found in marijuana that produces a ‘high’. Landis discovered it after life as a professional athlete left him with ‘numerous lingering painful injuries’. CBD, he explained, allowed him a route away from opioids and other addictive drugs that in the past doctors were quick to prescribe. He then created Colorado-based Floyd’s of Leadville to share with the world his experience of the advantages of CBD.
“For me it’s been beneficial for anxiety and to some extent pain as well,” he told FoodNavigator.
But the current lack of hard evidence to back up the many perceived health benefits is one bottleneck for the CBD industry. Studies continue to show it may prove to be an option for, among other things, managing anxiety, insomnia, inflammation, arthritis, and even HIV symptoms. But these studies are as yet inconclusive. With the exception of Epidiolex – the first and only prescription CBD medicine approved by the FDA in the US which is used to treat seizures – no…
[ad_2]
MMP News Author
Source link