[ad_1]
Women with breast cancer should include the “liberal culinary use of cruciferous vegetables.”
Both the Women’s Intervention Nutrition Study and the Women’s Health Initiative study showed that women randomized to a lower-fat diet enjoyed improved breast cancer survival. However, in the Women’s Healthy Eating and Living Study, women with breast cancer were also randomized to drop their fat intake down to 15 to 20 percent of calories, yet there was no difference in breast cancer relapse or death after seven years.
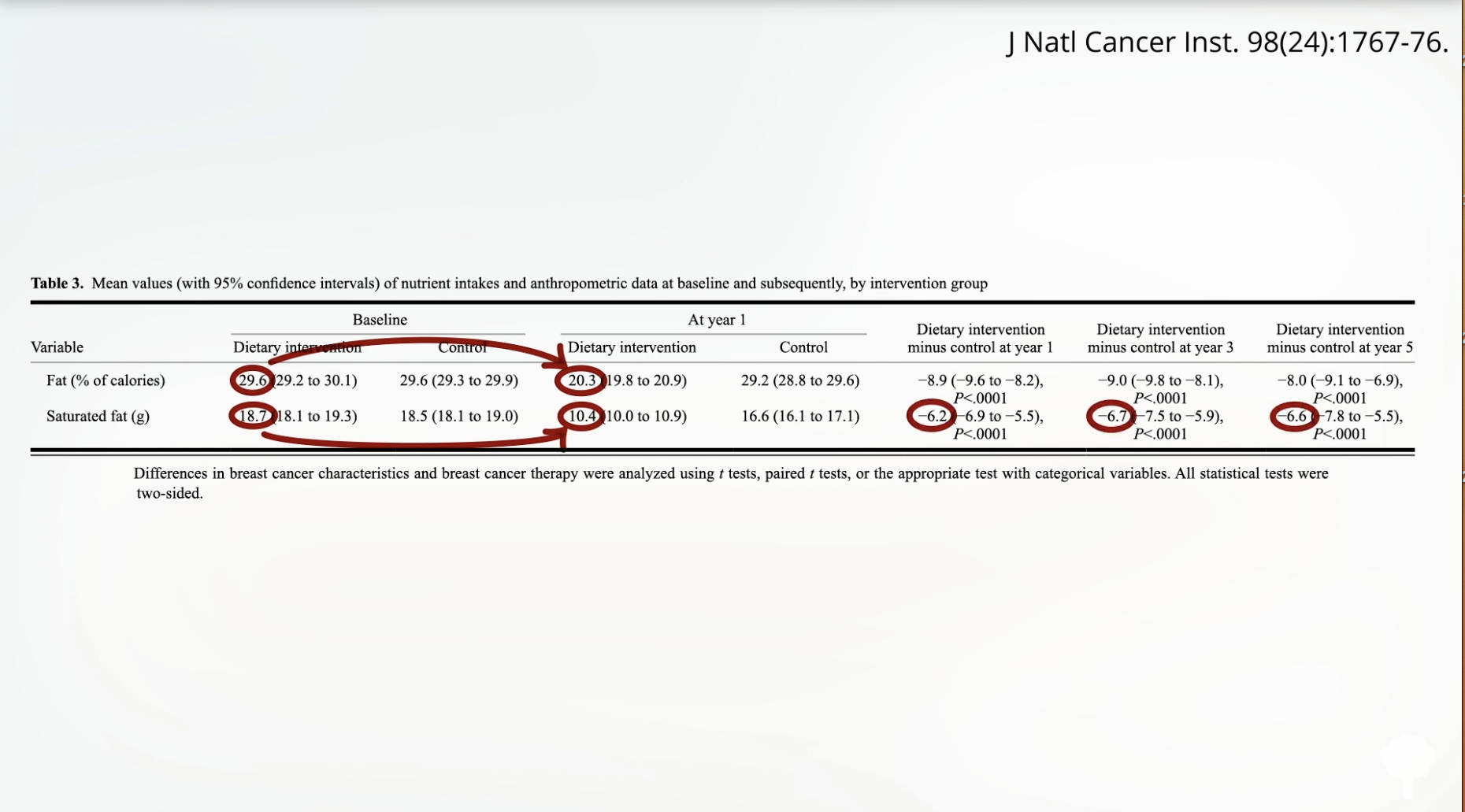
Any time there’s an unexpected result, you must question whether the participants actually followed through with study instructions. For instance, if you randomized people to stop smoking and they ended up with the same lung cancer rates as those in the group who weren’t instructed to quit, one likely explanation is that the group told to stop smoking didn’t actually stop. In the Women’s Healthy Eating and Living Study, both the dietary intervention group and the control group started out at about 30 percent of calories from fat. Then, the diet group was told to lower their fat intake to 15 to 20 percent of calories. By the end of the study, they had in fact gone from 28.5 percent fat to 28.9 percent fat, as you can see below and at 1:16 in my video The Food That Can Downregulate a Metastatic Cancer Gene. They didn’t even reduce their fat intake. No wonder they didn’t experience any breast cancer benefit.
When you put together all the trials on the effect of lower-fat diets on breast cancer survival, even including that flawed study, you see a reduced risk of breast cancer relapse and a reduced risk of death. In conclusion, going on a low-fat diet after a breast cancer diagnosis “can improve breast cancer survival by reducing the risk of recurrence.” We may now know why: by targeting metastasis-initiating cancer cells through the fat receptor CD36.
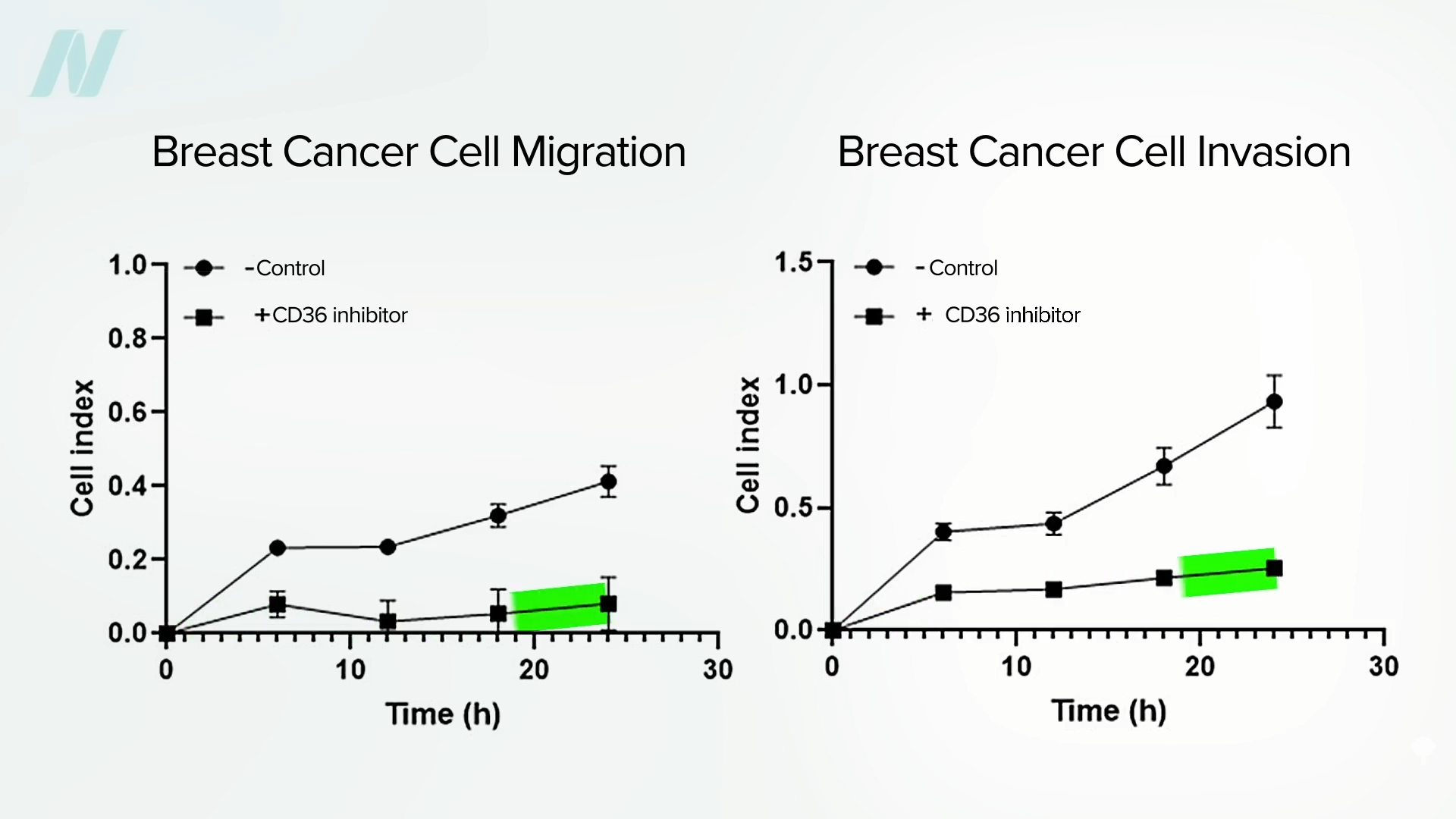
We know that the cancer-spreading receptor is upregulated by saturated fat. Is there anything in our diet that can downregulate it? Broccoli.
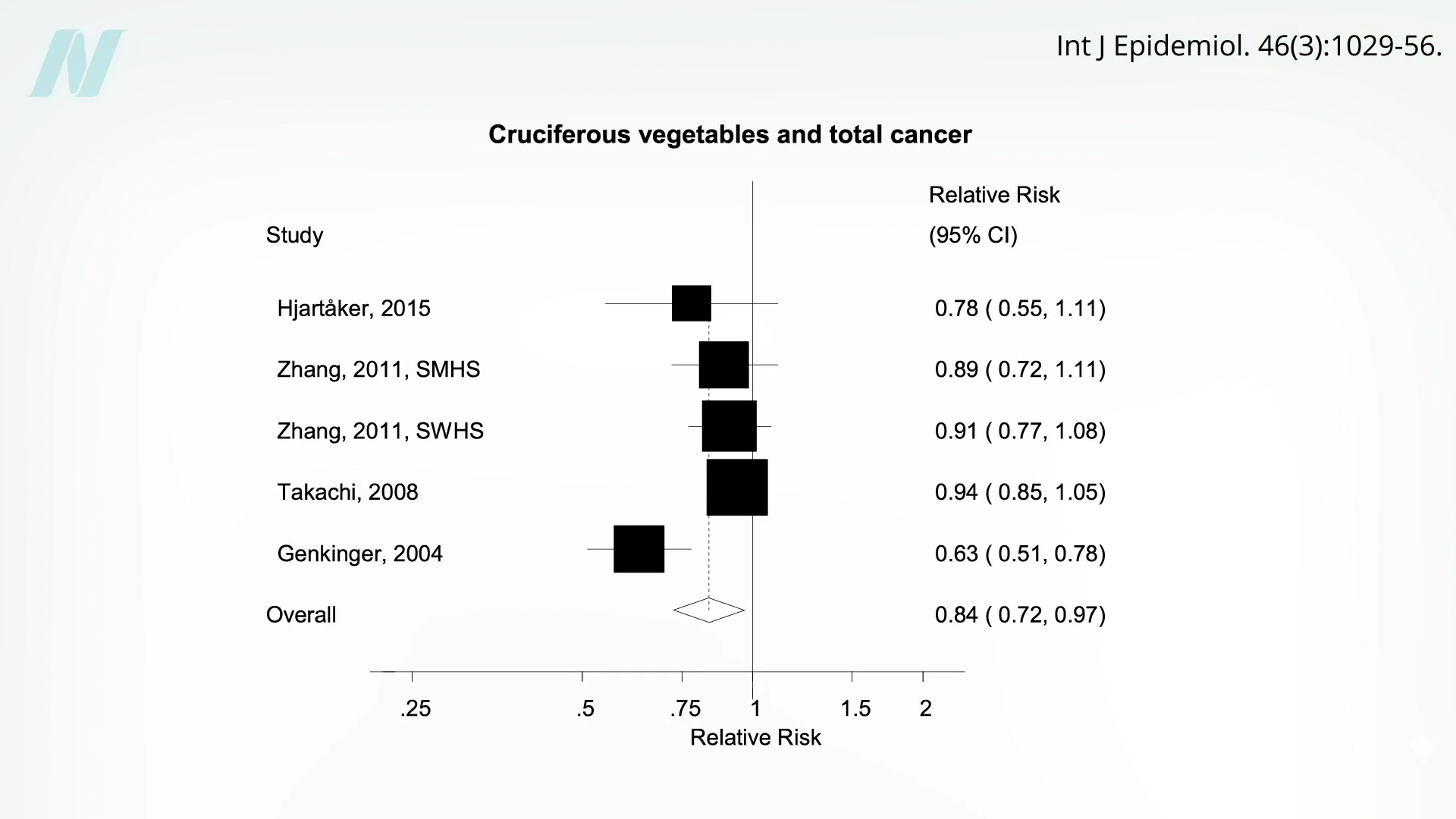
Broccoli appears to decrease CD36 expression by as much as 35 percent (in mice). Of all fruits and vegetables, cruciferous vegetables like broccoli were the only ones associated with significantly less total risk of cancer and not just getting cancer in the first place, as you can see here and at 2:19 in my video.
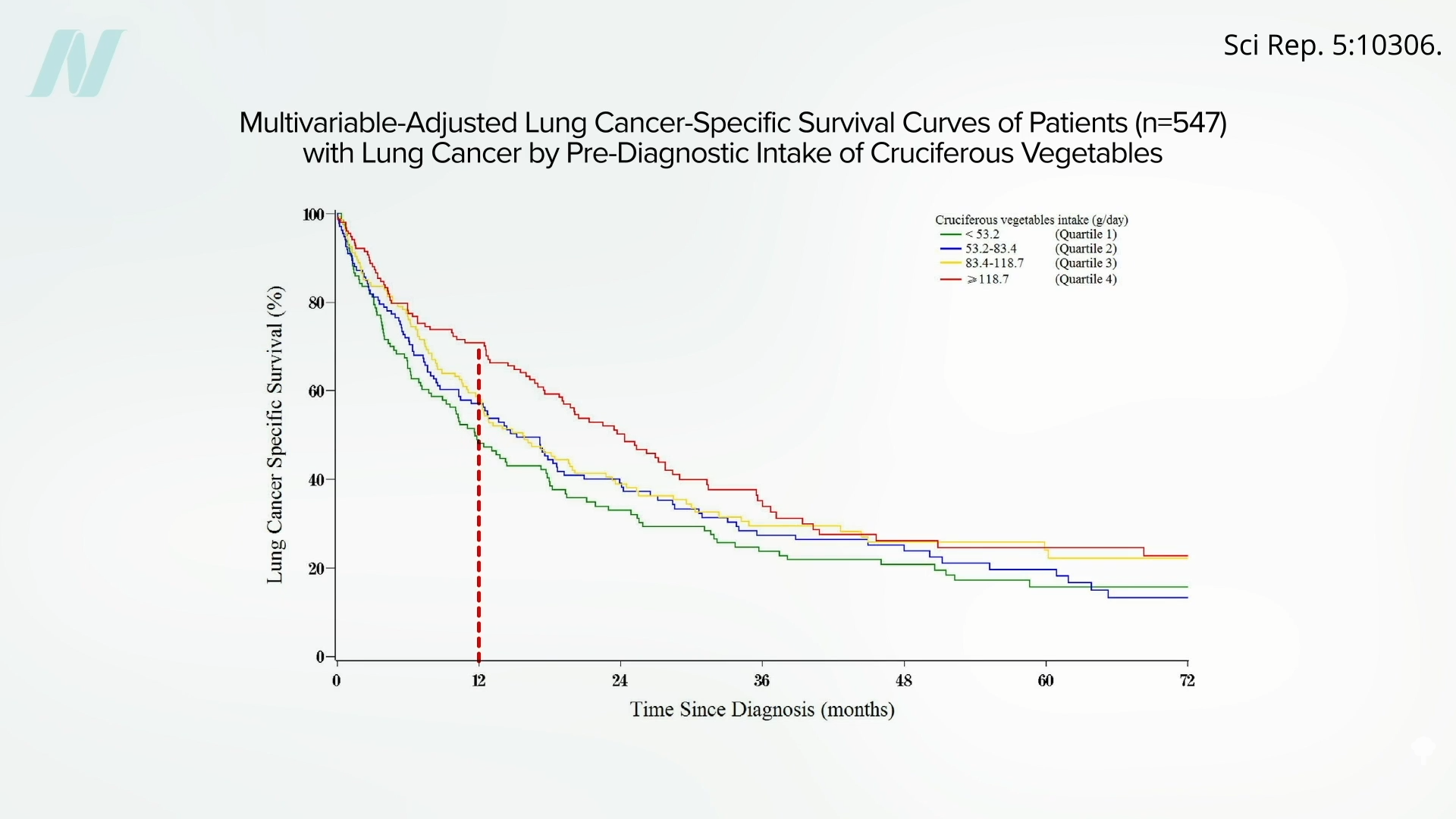
Those with bladder cancer who eat broccoli also appear to live longer than those who don’t, and those with lung cancer who eat more cruciferous veggies appear to survive longer, too.
For example, as you can see below and at 2:45 in my video, one year out, about 75 percent of lung cancer patients eating more than one serving of cruciferous vegetables a day were still alive (the top line in red), whereas, by then, most who had been getting less than half a serving a day had already died from their cancer (the bottom line in green).
Ovarian cancer, too. Intake of cruciferous vegetables “significantly favored survival,” whereas “a survival disadvantage was shown for meats.” Milk also appeared to double the risk of dying. Below and at 3:21 in my video are the survival graphs. Eight years out, about 40 percent of ovarian cancer patients who averaged meat or milk every day were deceased (the boldest line, on the bottom), compared to only about 20 percent who had meat or milk only a few times a week at most (the faintest line, on the top).
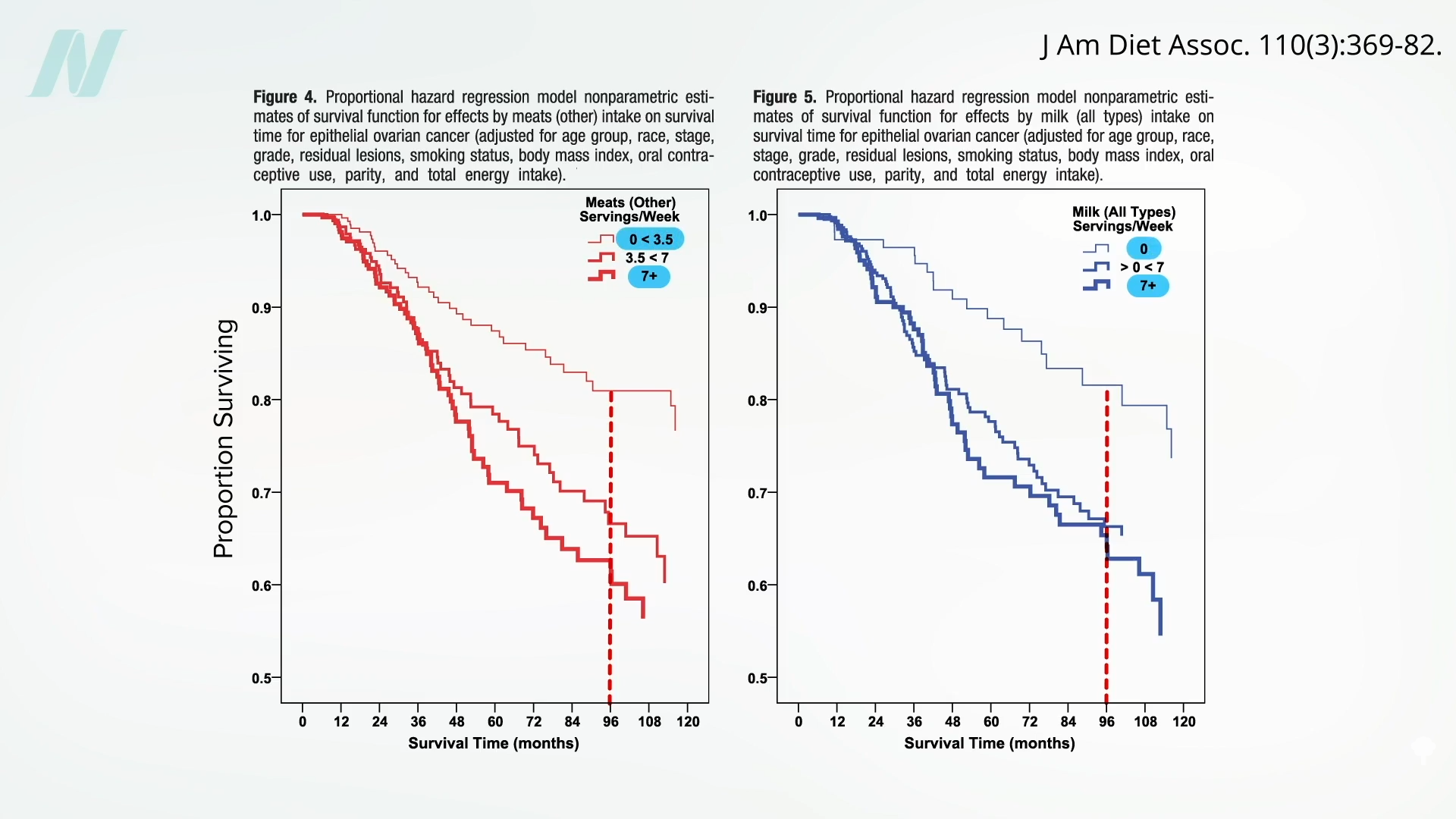
Now, it could be that the fat and cholesterol in meat increased circulating estrogen levels, or it could be because of meat’s growth hormones or all its carcinogens. And galactose, the sugar naturally found in milk, may be directly toxic to the ovary. Dairy has all its hormones, too. However, the lowering of risk with broccoli and the increasing of risk with meat and dairy are also consistent with the CD36 mechanism of cancer spread.
Researchers put it to the test in patients with advanced pancreatic cancer who were given pulverized broccoli sprouts or a placebo. The average death rate was lower in the broccoli sprout group compared to the placebo group. After a month, 18 percent of the placebo group had died, but none in the broccoli group. By three months, another 25 percent of the placebo group had died, but still not a single death in the broccoli group. And by six months, 43 percent of the remaining patients in the placebo group were deceased, along with the first 25 percent of the broccoli group. Unfortunately, even though the capsules for both groups looked the same, “true blinding was not possible,” and the patients knew which group they were in “because the pulverized broccoli sprouts could be easily distinguished from the methylcellulose [placebo] through their characteristic smell and taste.” So, we can’t discount the placebo effect. What’s more, the study participants weren’t properly randomized “because many of the patients refused to participate unless they were placed into the [active] treatment group.” That’s understandable, but it makes for a less rigorous result. A little broccoli can’t hurt, though, and it may help. It’s the lack of downsides of broccoli consumption that leads to “Advising Women Undergoing Treatment for Breast Cancer” to include the “liberal culinary use of cruciferous vegetables,” for example.
It’s the same for reducing saturated fat. The title of an editorial in a journal of the National Cancer Institute asked: “Is It Time to Give Breast Cancer Patients a Prescription for a Low-Fat Diet?” “Although counseling women to consume a healthy diet after breast cancer diagnosis is certainly warranted for general health, the existing data still fall a bit short of proving this will help reduce the risk of breast cancer recurrence and mortality.” But what do we have to lose? After all, it’s still certainly warranted for general health.
[ad_2]
Michael Greger M.D. FACLM
Source link