[ad_1]
Let’s talk about treating weight loss, cholesterol, and PCOS with diet. What can an eighth of a teaspoon a day of onion powder do for body fat, and what can raw red onion do for cholesterol?
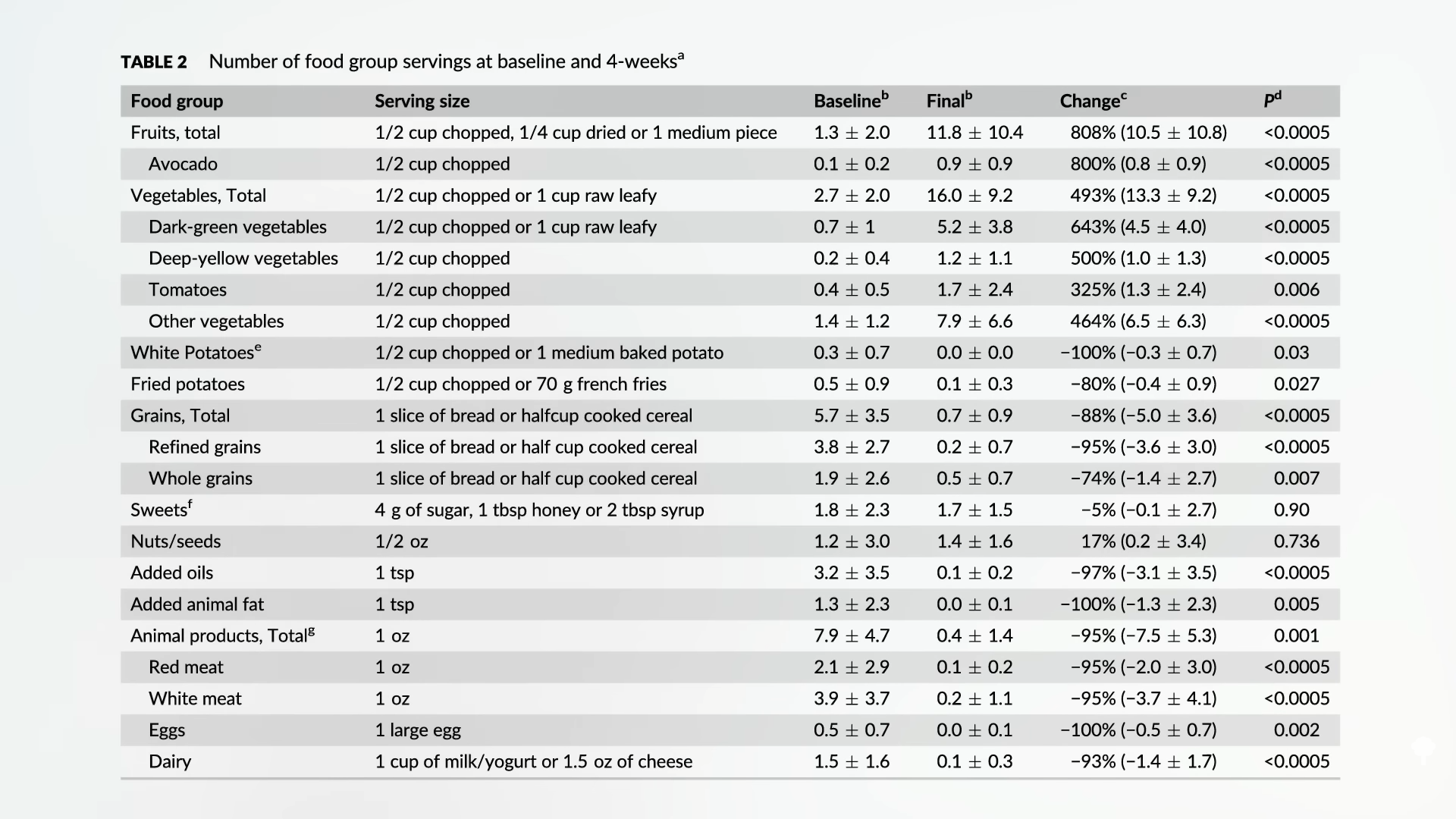
In one of my previous videos about onions, I talked about the data supporting—or not supporting—the role of onions in boosting testosterone in men, protecting bone health, controlling allergies, and dealing with the side effects of chemotherapy. What about weight loss? Enter the “Effect of Steamed Onion (ONIRO) Consumption on Body Fat and Metabolic Profiles in Overweight Subjects.” Researchers used steamed onions, which aren’t as spicy and have a weaker smell, so they could better disguise them as a placebo. They dried them into onion powder and gave people a minuscule amount—about an eighth of a teaspoon (300 mg) a day. Surely, a little daily dusting of onion powder wouldn’t affect people’s weight. But check out the results reported in the abstract: Measurements using a DEXA scan showed a significant reduction in body fat mass, and a CT scan revealed a significant decrease in whole, visceral, and subcutaneous fat areas.
Hold on. If a little onion powder is so effective for weight loss, why wasn’t it featured in my book How Not to Diet? Because, as so often happens in studies, the spin in the abstract doesn’t accurately represent the actual data. The DEXA scan results measured no significant change of fat in the group that got the placebo capsules. They only appeared to lose about a spoonful (7 g) of fat, whereas the group unknowingly taking an eighth of a teaspoon of onion powder stuffed into capsules lost nearly one and a half pounds (0.64 kg) of body fat—a significant drop from baseline, but not a statistically significant drop compared to the placebo group, meaning the loss could have just been due to chance. Same thing with the CT scan results: 5 times more loss of overall fat and over 30 times more loss of the dangerous visceral fat, but the results did not reach statistical significance compared to placebo.
A more recent study tried four teaspoons (9 g) of onion powder a day and similarly failed to accelerate the loss of visceral, total, or subcutaneous fat compared to placebo—but the placebo was also four teaspoons (9 g) of onion powder a day. They used yellow onions versus white onions, and it seems they both may have caused a loss of abdominal body fat, without a significant difference between them. Either way, you might look at these two studies and think, sure, but what are the downsides? It’s only an eighth of a teaspoon of onion powder a day, so why not give it a try? It can’t hurt, but we just don’t have enough evidence to be confident it will actually help.
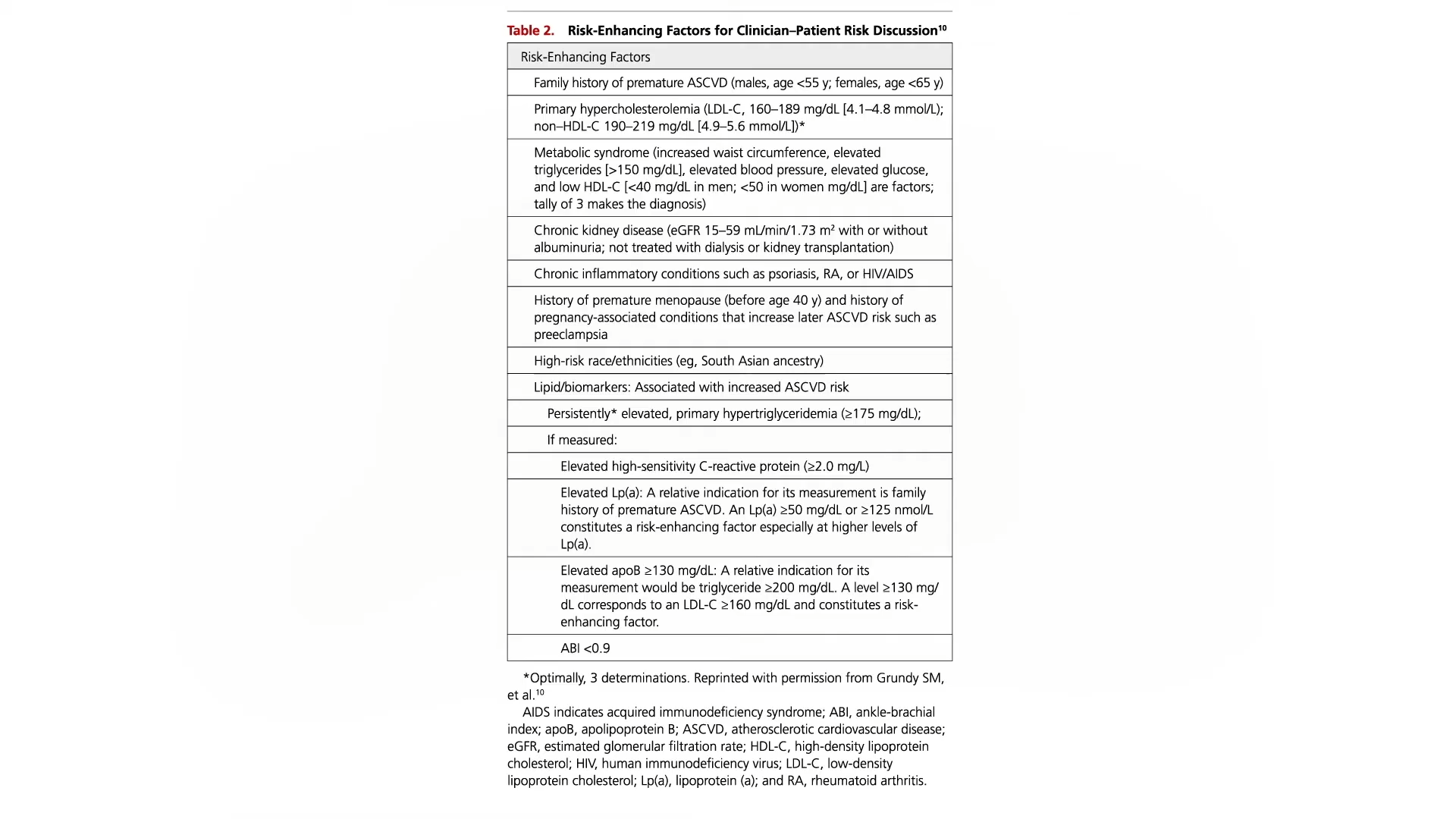
Let’s talk about polycystic ovary syndrome, also known as PCOS. It’s one of the most common hormone disorders, affecting 5% to 10% of reproductive-aged women. In addition to causing symptoms like irregular periods, “PCOS is a pre-diabetic state, with decreased insulin sensitivity.” PCOS treatment is challenging due to medication side effects. So, are there dietary options? How about a randomized controlled clinical trial of raw red onion intake?
Why onions? Well, onion extracts can evidently improve blood sugar and insulin sensitivity in rats with diabetes and, more importantly, were found to reduce blood sugar levels in humans with diabetes, but evidently not in non-diabetic humans. People with PCOS are kind of pre-diabetic, so would it work for them? First, let’s look at those other two studies. To study the “Metabolic Effects of Onion and Green Beans,” people with diabetes spent a week eating either a small onion (60 g) each day or the same diet with about six cups (600 g) of green beans instead—and both approaches worked. The onion lowered people’s blood sugar levels by about 10% compared to a non-onion control diet, while the green beans lowered them by roughly 15% compared to the control.
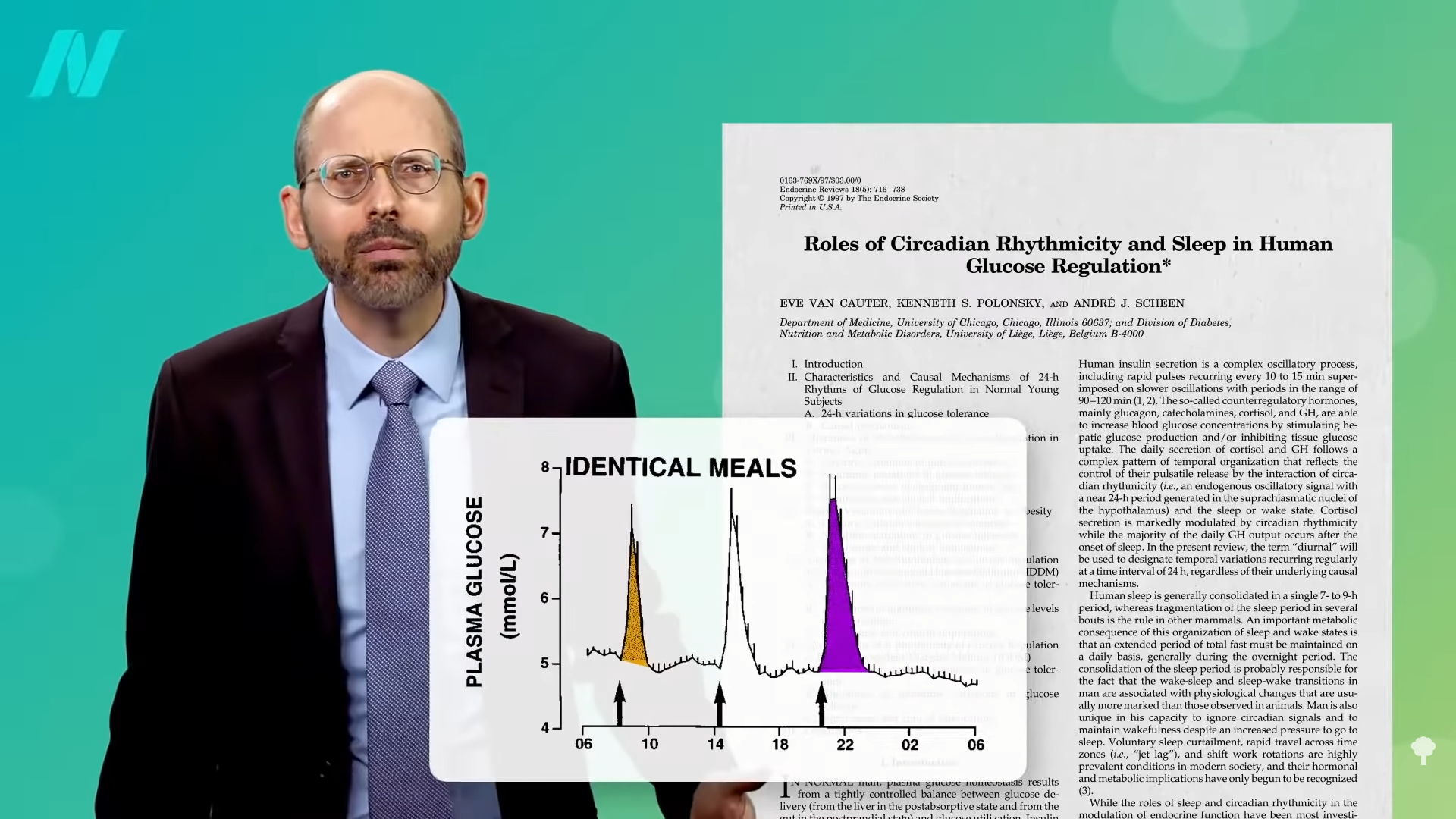
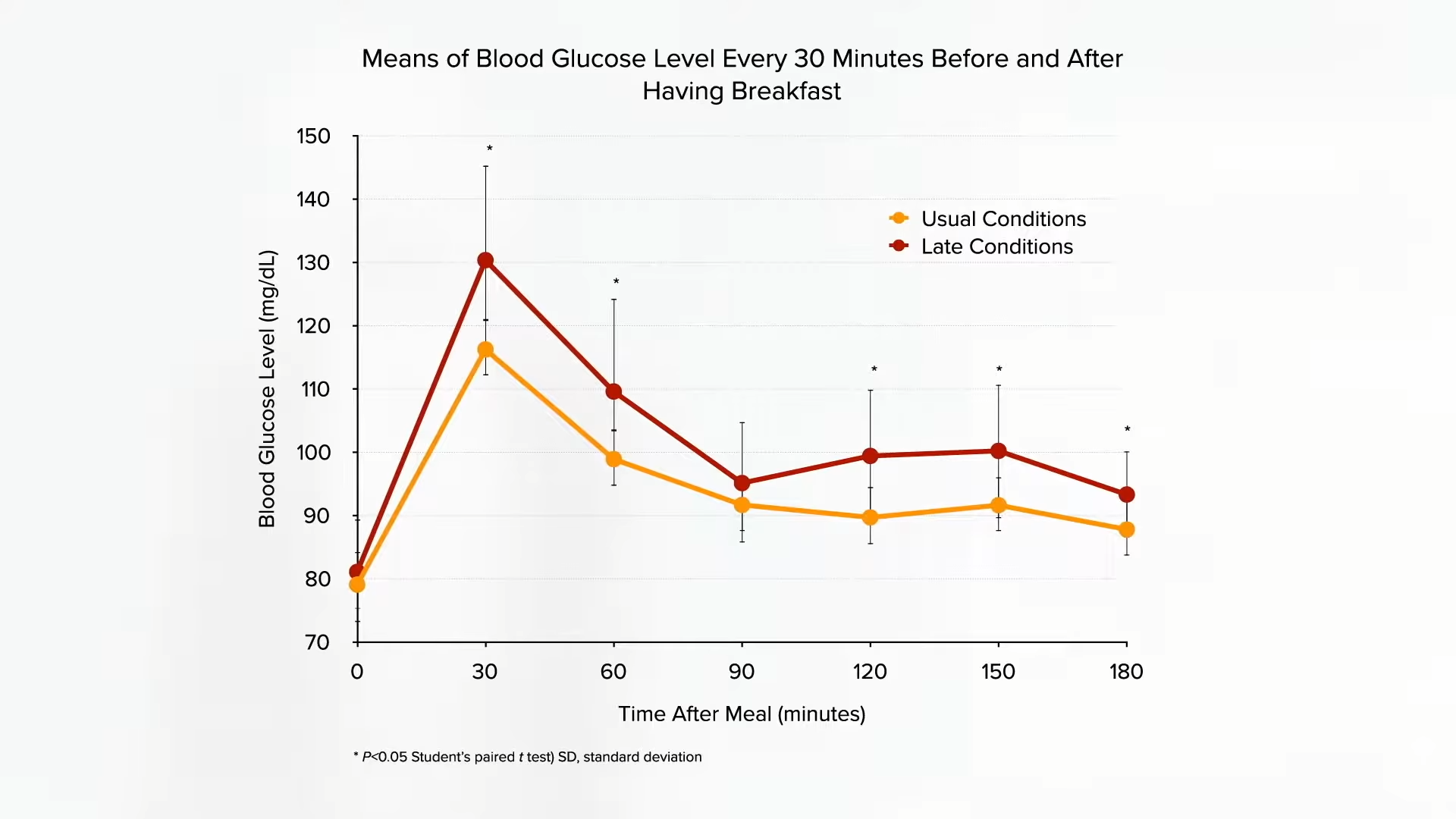
Here’s the study that supposedly shows no blood sugar benefits for people without diabetes. It’s true—onions don’t seem to lower normal blood sugar levels, which is a good thing, but check out what happens when you feed people sugar. Have people consume about two and a half tablespoons (50 g) of corn syrup, and their blood sugar levels shoot up over the next two hours before their body can tamp it back down. But give people the exact same amount of sugar along with more and more onion extract, and the blood sugar spike is significantly dampened, almost as much as if you had instead given them an antidiabetic drug, as you can see below and at 4:00 in my video Onions Put to the Test for Weight Loss, Cholesterol, and PCOS Treatment.
We see the same blunting effect on blood sugar when people get a shot of adrenaline and eat onion extract, compared to receiving adrenaline without the onion extract, as you can see below and at 4:11 in my video.
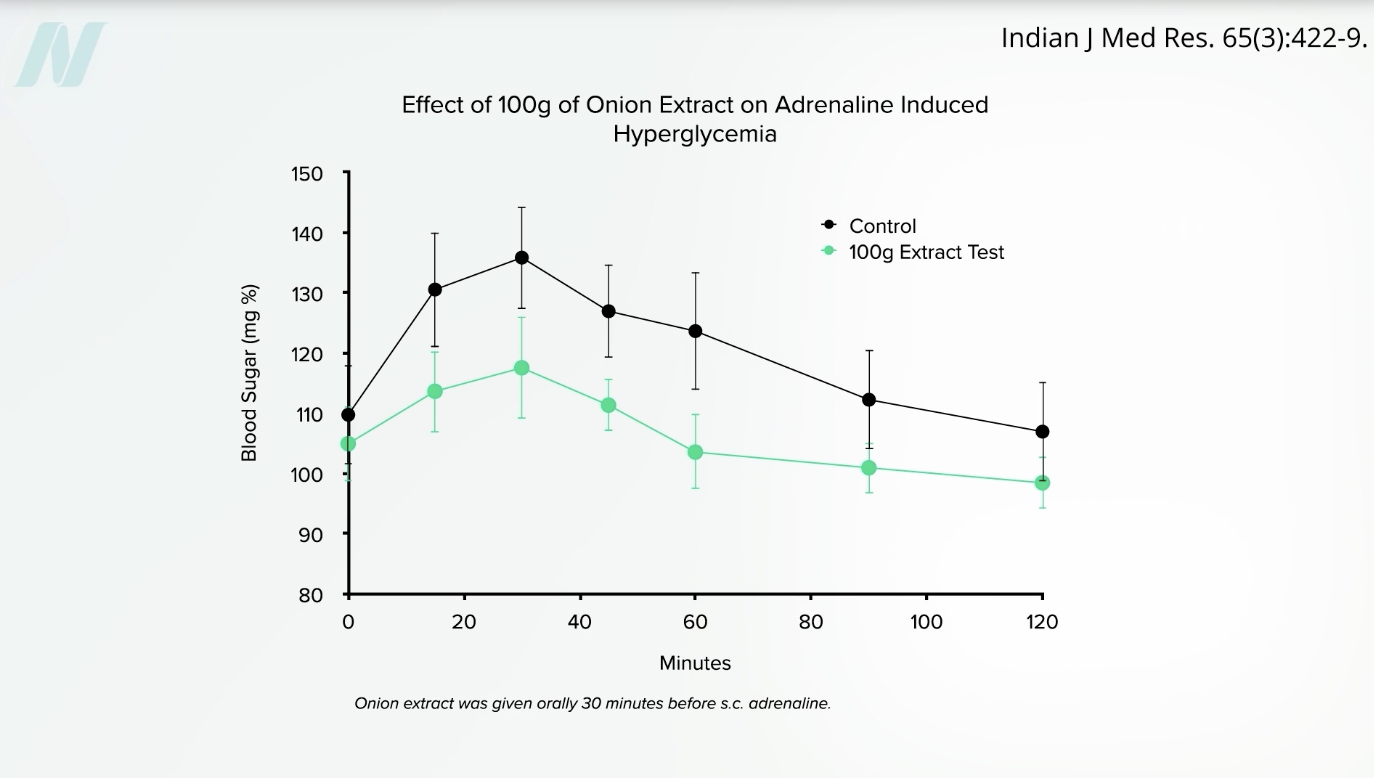
So, are there blood sugar benefits for both people with and without diabetes? No difference was found in blood sugar levels or other markers of insulin resistance between the high-onion and low-onion groups of PCOS patients, nor were there any differences in a marker of inflammation between the two groups. But women with PCOS aren’t just at higher risk for diabetes and inflammation—they are also at higher risk for high cholesterol.
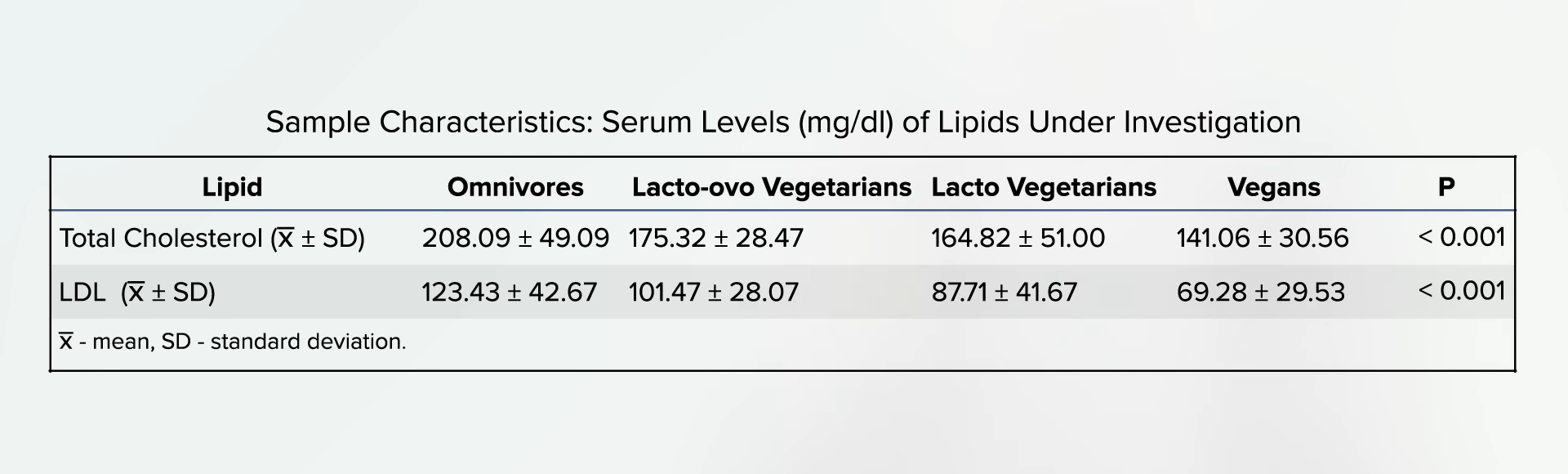
Women with PCOS are over seven times more likely to have a heart attack and develop heart disease, the number one killer of women. But consuming raw red onion appears to be effective in lowering cholesterol, though the group that ate more onions only dropped their LDL cholesterol about 5 points (5 mg/dL), which was not significantly different than the group that ate fewer onions.
I did find this study from 50 years ago where researchers fed people nearly an entire stick (100 g) of butter, and their cholesterol shot up about 30 points within hours of consumption but by only 9 points or 3 points when combined with about a third of a cup (50 g) of raw or boiled onion. The moral of the story: Don’t eat a stick of butter.
Doctor’s Note
Check out the previous video I mentioned: Friday Favorites: Are Onions Beneficial for Testosterone, Osteoporosis, Allergies, and Cancer?.
[ad_2]
Michael Greger M.D. FACLM
Source link


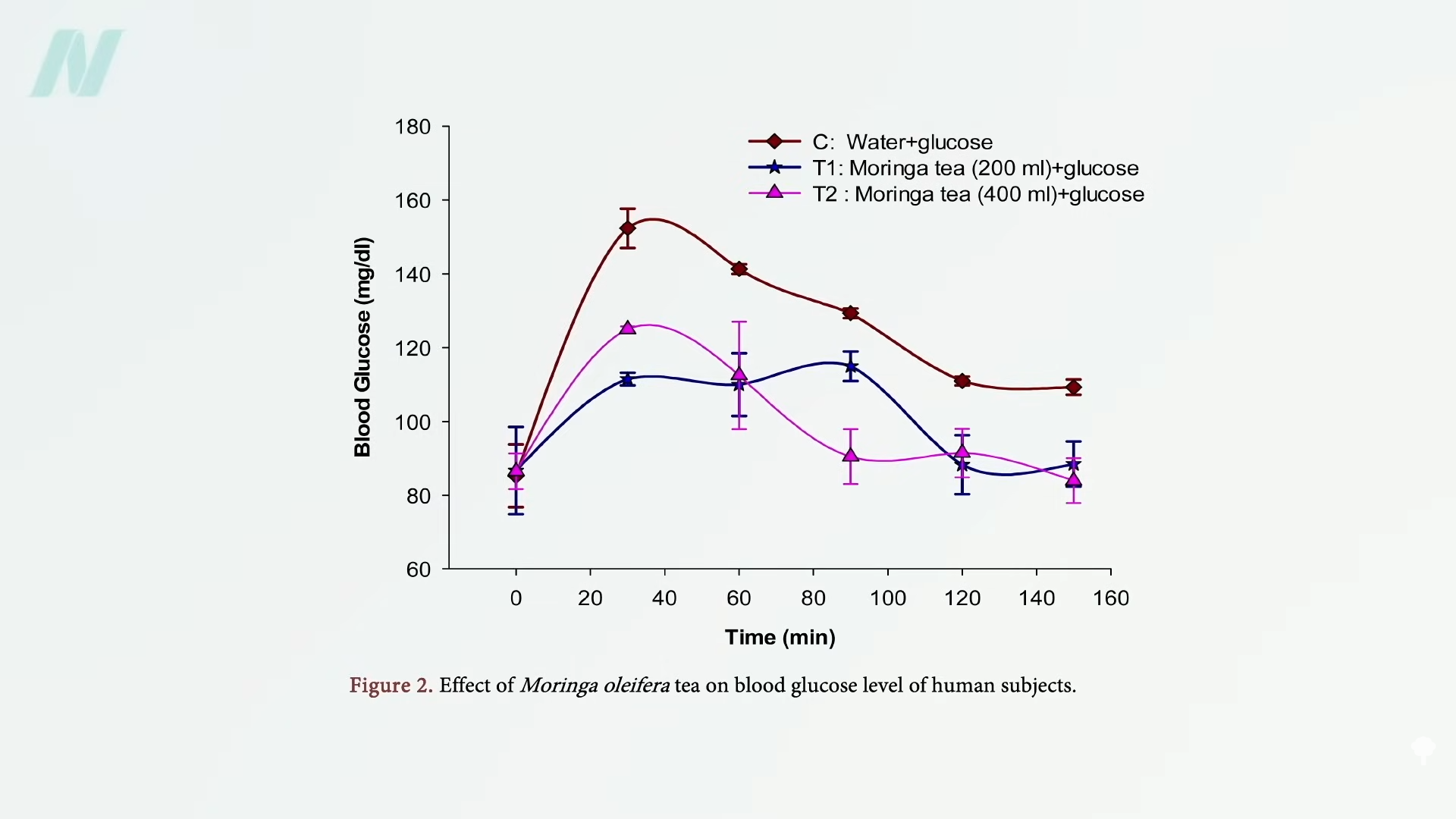 People with diabetes were
People with diabetes were