[ad_1]
Coronary artery disease, the number one killer of men and women, involves blockages in the blood vessels that supply the heart muscle. As discussed in my video Do Angioplasty Heart Stent Procedures Work?, low blood flow can lead to a type of chest pain called angina or, if severe enough, a heart attack. Plant-based diets and lifestyle programs have been shown to help reverse these blockages by treating the cause of why our arteries are clogging up in the first place. But, for those unable or unwilling to change their diets, there are drugs that may help, as well as more invasive surgical treatments.
What Is a Heart Stent?
You may have heard of open-heart surgery, performed to try to bypass the blockage, or percutaneous coronary intervention. As discussed in my video Why Angioplasty Heart Stents Don’t Work Better, historically, the more common procedure was angioplasty, wherein a tiny balloon is inserted into a narrowed coronary artery feeding your heart to force it to open wider to improve blood flow. Then, stents came into vogue. Instead of just ballooning up the artery, how about permanently inserting a metal mesh tube to prop open the artery? Stents are typically inserted in the groin and threaded all the way up into the heart, and, while stents used to be mostly bare metal, there are now fancy new drug-eluting stents that not only force open arteries, but they also slowly release pharmaceuticals.
How Serious Is Having a Stent Put In?
The surgical procedure carries risks—including death. In an emergency setting, while you’re actively having a heart attack, angioplasty can be lifesaving, but hundreds of thousands of these procedures are for stable coronary artery disease, for which there appears to be little or no benefits. As discussed in my video The Risks of Heart Stents, doctors appear to be killing or stroking out thousands of people a year for nothing, and that isn’t even counting the tens of thousands of silent mini-strokes caused by these procedures that may contribute to cognitive decline. Indeed, 11 to 17 percent of people who go through angioplasty or stenting come away with new brain lesions—up to one in six patients.
Do Stents Work?
Angioplasty and stents for non-emergency coronary artery disease are among the most common invasive procedures performed in the United States. Millions of people have gotten stents for stable coronary artery disease, yet it now appears that for such patients, angioplasty and stent placement do not actually prevent heart attacks, do not offer long-term angina pain relief, and do not improve survival. Why? Because the most dangerous plaques—the ones most vulnerable to rupture leading to a heart attack—are not the ones doctors put stents into. They often aren’t the ones that are even seen on angiogram to be obstructing blood flow.
Indeed, in 2007, we learned from the COURAGE trial that angioplasty and stents don’t reduce the risk of death or heart attack, but patients didn’t seem to get the memo. As discussed in my video Why Are Stents Still Used If They Don’t Work?, only 1 percent realize there was no mortality or heart attack benefit, perhaps because most cardiologists failed to happen to mention that fact. One can imagine that if patients actually understood all they were getting was symptomatic relief, they’d be less likely to go under the knife. Ten years later, the ORBITA trial was published, showing even the promise of symptom relief was an illusion.
Are Stents Really Necessary?
The implications are profound and far-reaching. First and foremost, the results showed unequivocally that there are no benefits to non-emergency angioplasty and stents for stable heart disease. Basically, patients would be risking harm for no benefit whatsoever, so it’s hard to imagine a scenario where a fully-informed patient would choose an invasive procedure for nothing.
Yet angioplasty and stent placement continue to be frequently performed for patients with non-emergency coronary artery disease, despite clear evidence that it provides minimal benefit, as discussed in my video Angioplasty Heart Stent Risks vs. Benefits. For example, it does not prevent heart attacks or death, yet as many as nine out of ten patients mistakenly believed that the procedure would reduce their chances of having a heart attack.
What Are the Side Effects of Heart Stents?
Stent placement and the blood-thinner drugs you have to go on after the surgery can cause complications, including heart failure, stroke, and death. The risks are relatively low; there’s less than a 1 percent chance it will kill you or stroke you out. The 15 percent risk of heart attack is only if your stent clogs at a later date, which only happens about 1 percent of the time in the near-term. There is a 13 percent risk of kidney injury, due to the dyes that must be injected, but that typically heals on its own. The most serious complications—including death—only happen in about 1 in 150 cases. However, you have to multiply that by the fact that hundreds of thousands of these procedures are performed every year.
And, again, although stents appeared to offer immediate relief of angina chest pain in stable patients with coronary artery disease, they do not offer long-term angina pain relief and they didn’t actually translate into lower risk of heart attack or death. More on this in my video Do Heart Stent Procedures Work for Angina Chest Pain?.
Diet After Heart Attack and Stents
Should we be surprised that angioplasty and stents fail to improve prognosis? After all, neither does anything to modify the underlying disease process itself. In other words, they don’t treat the cause. As discussed in my video Heart Stents and Upcoding: How Cardiologists Game the System, even if stents helped with symptoms beyond the placebo effect, they would still just be treating the symptoms, not the disease, so it’s no wonder the disease continues to progress until the patient is disabled into death.
Thankfully, we are on the cusp of a seismic revolution in health: not another pill, procedure, or operation, but, instead, treating the underlying cause of heart disease with whole food, plant-based nutrition, the mightiest tool medicine has ever had in its toolbox.
Heart-Healthy Eating
The most likely reason the majority of our loved ones will die is heart disease. Atherosclerosis, or hardening of the arteries, begins in childhood, as discussed in my video How Not to Die from Heart Disease. The arteries of nearly all kids raised on the standard American diet already have fatty streaks marking the first stage of the disease—by the time they are ten years old. After that, the plaques start forming in our 20s, get worse in our 30s, and can then start killing us off. In our heart, it’s called a heart attack; in our brain, it can manifest as a stroke. So, for anyone reading this who is older than ten, the choice isn’t whether or not to eat healthfully to prevent heart disease—it’s whether or not you want to reverse the heart disease you likely already have.
Is that even possible? When researchers took people with heart disease and put them on the kind of plant-based diet followed by populations who did not get epidemic heart disease, their hope was that it might slow down the disease process or maybe even stop it. Instead, something miraculous happened. The disease actually started to reverse. It started to get better. As soon as patients stopped eating artery-clogging diets, their bodies were able to start dissolving away some of the plaque, opening up arteries without drugs and without surgery, suggesting their bodies wanted to heal all along, but just were never given the chance. That improvement in blood flow to the heart muscle itself was after just three weeks of eating healthfully.
Plant-based diets aren’t just safer and cheaper. They can work better because they let us treat the actual cause of the disease.
[ad_2]
Michael Greger M.D. FACLM
Source link








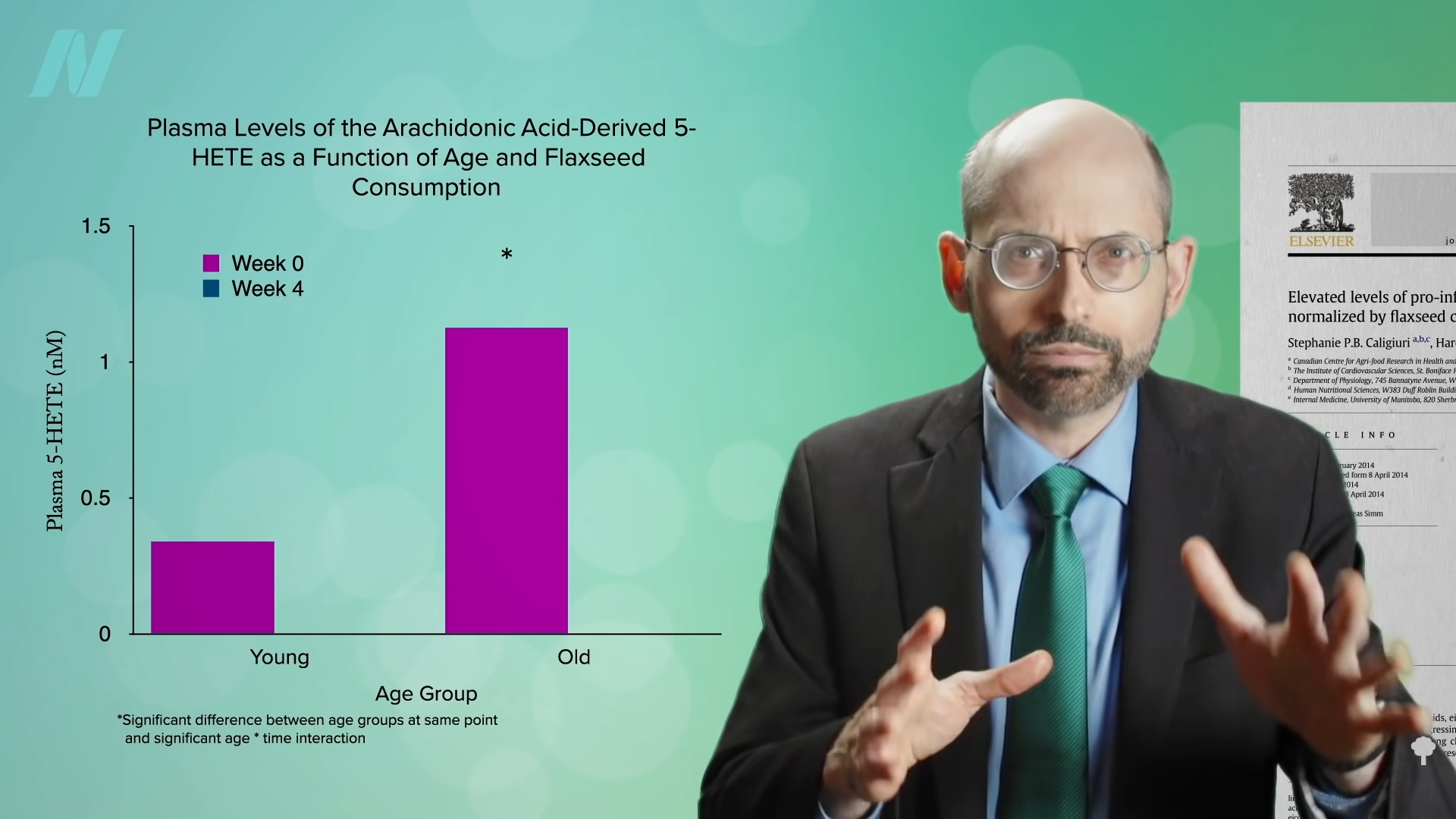
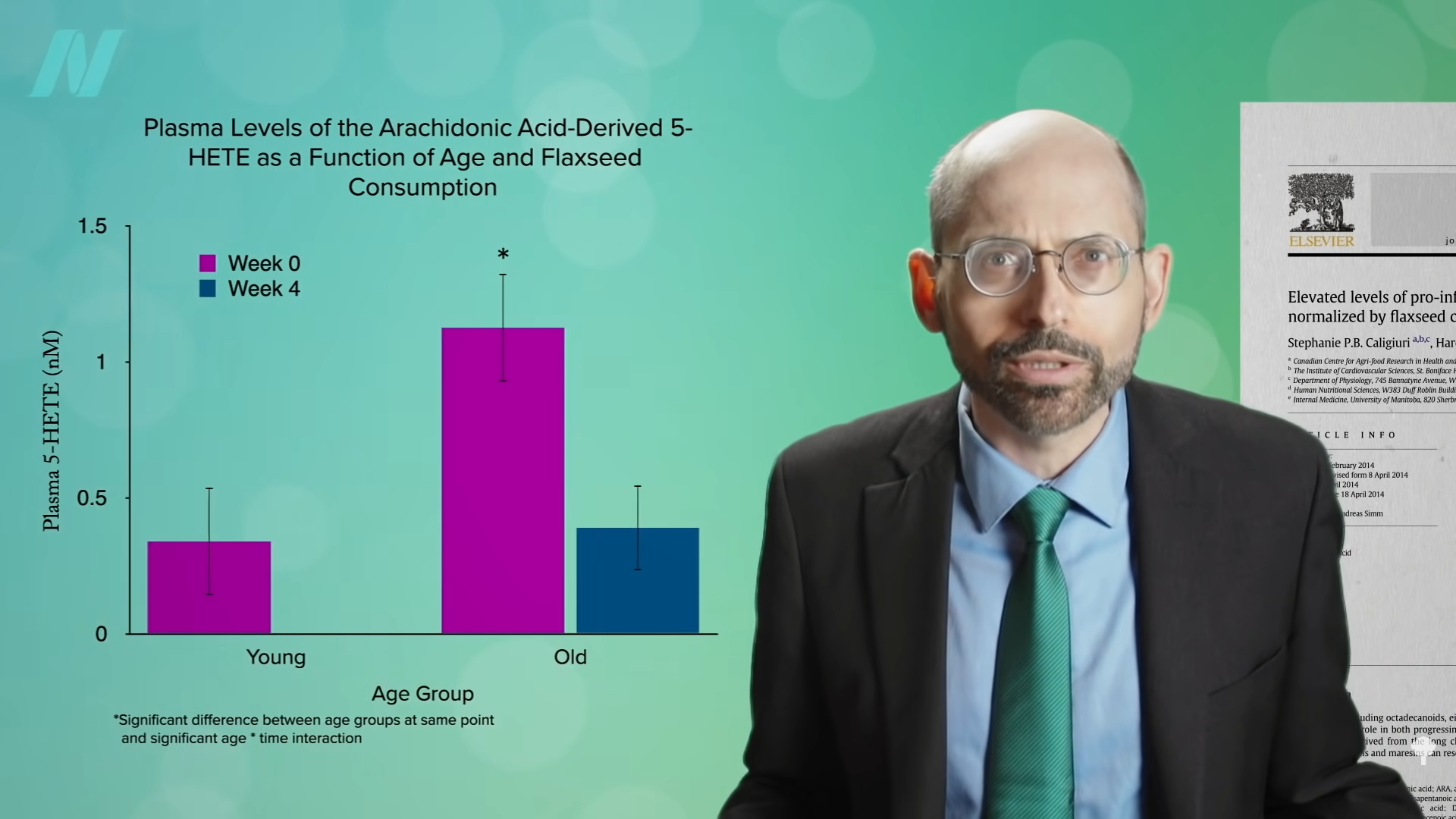



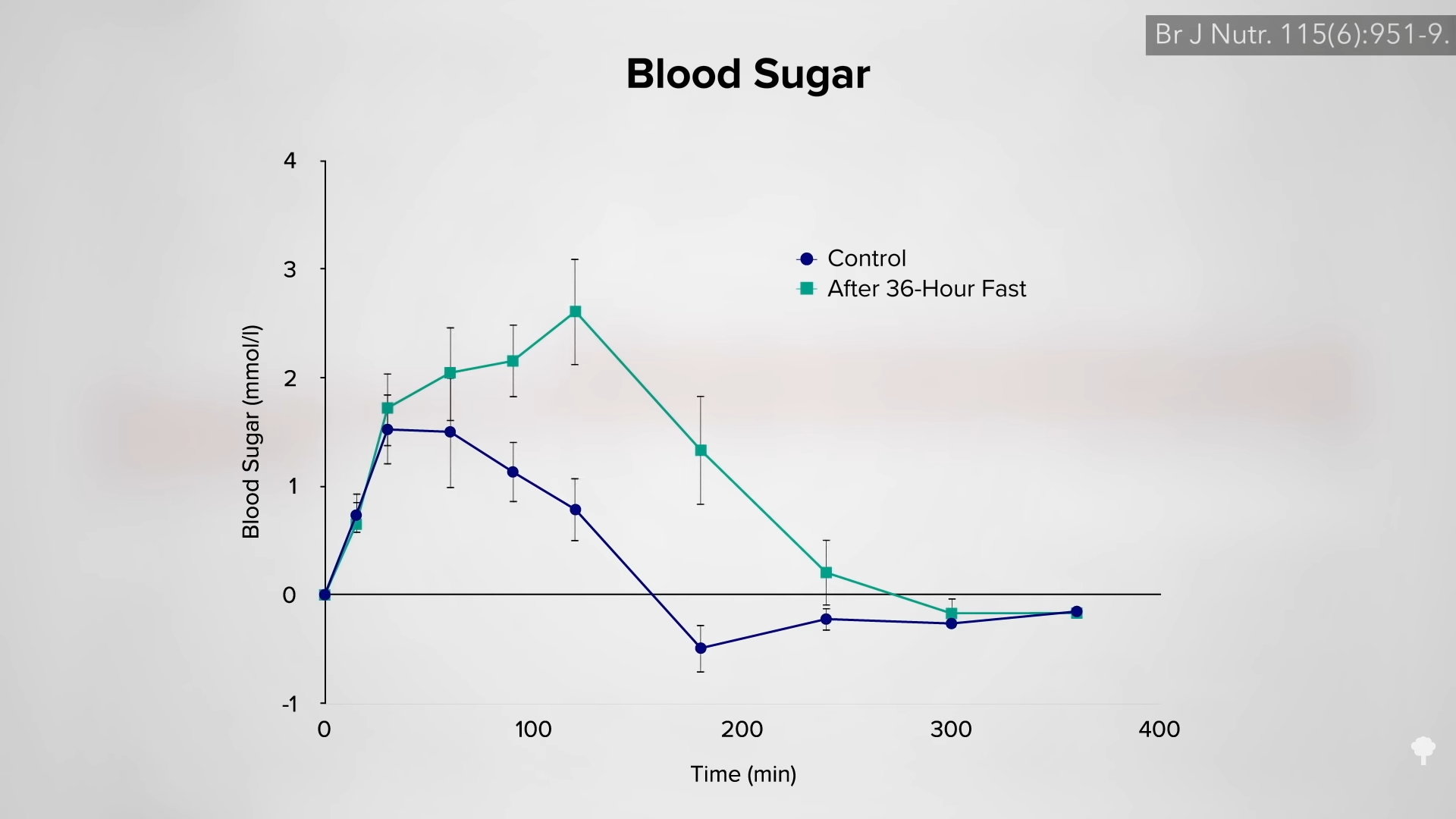
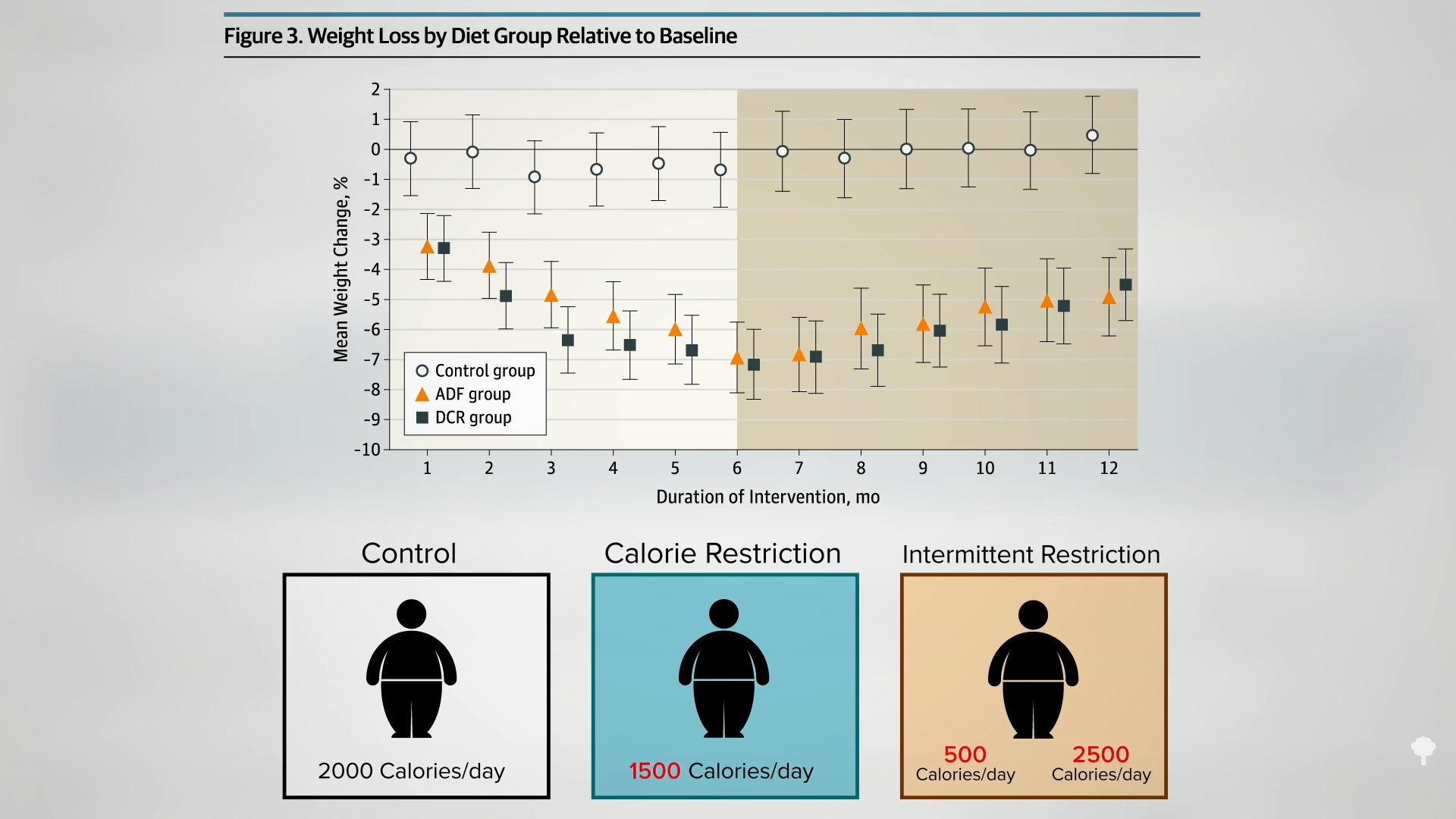
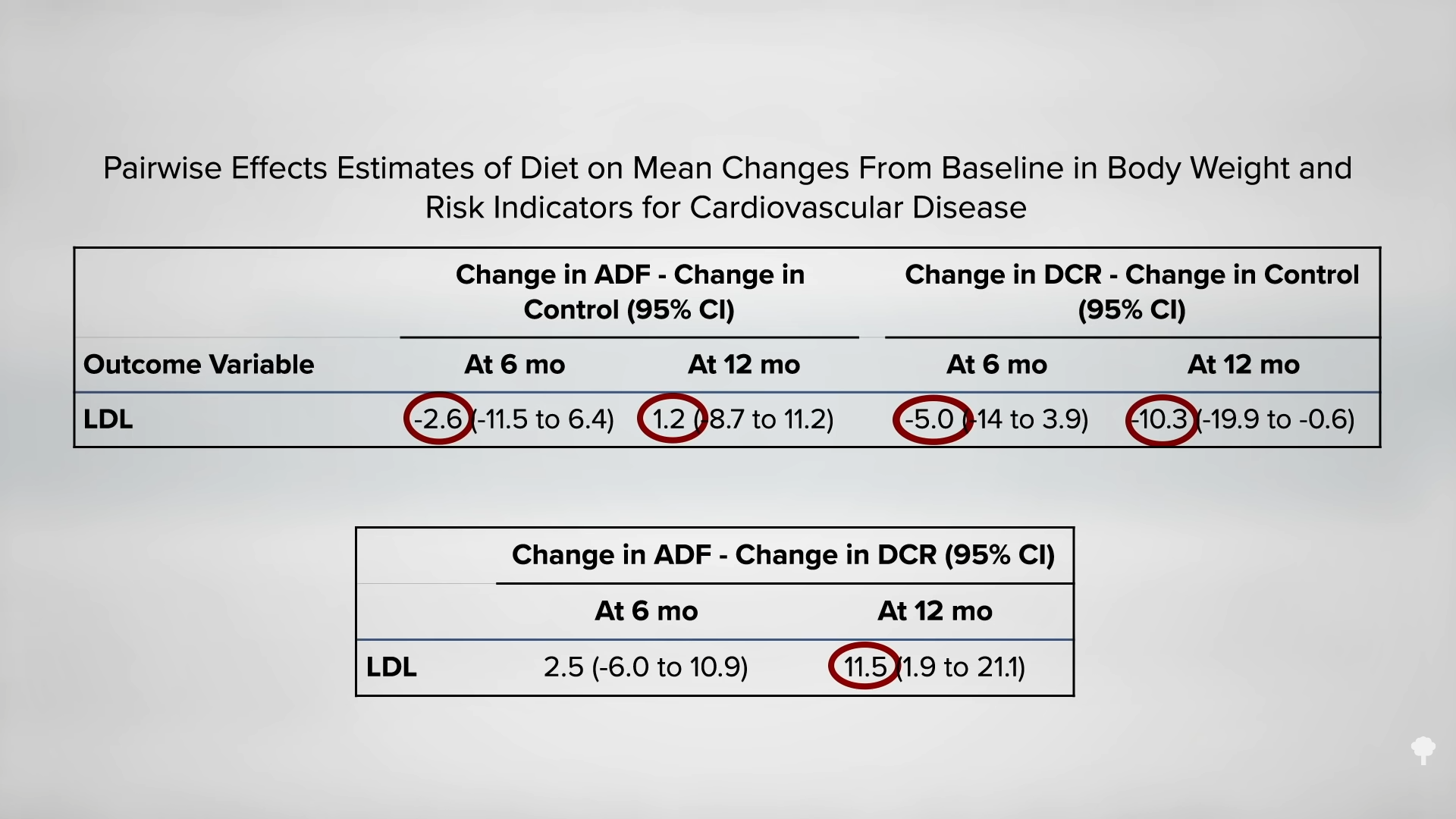

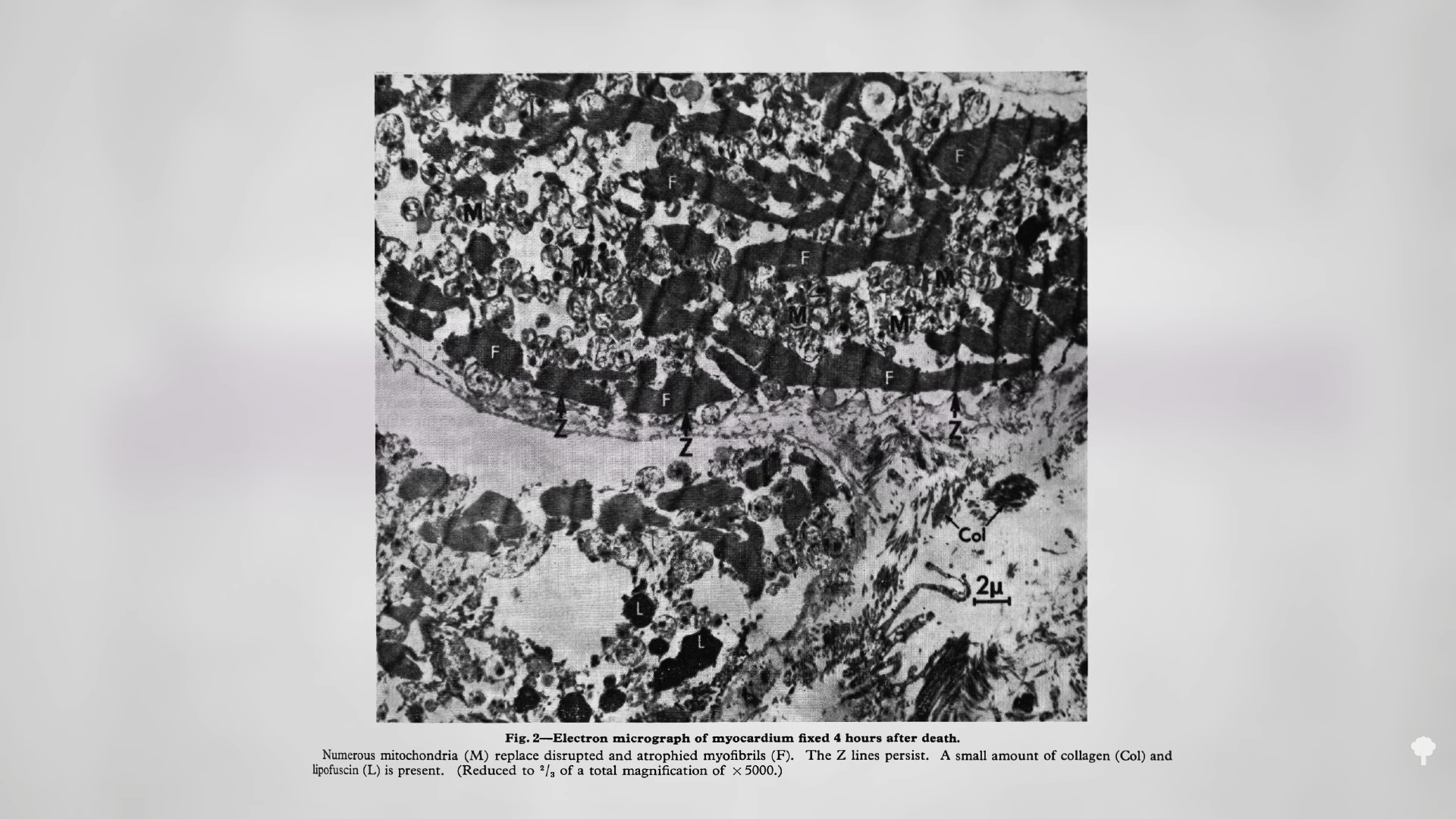
 Medically-supervised fasting has
Medically-supervised fasting has “
“
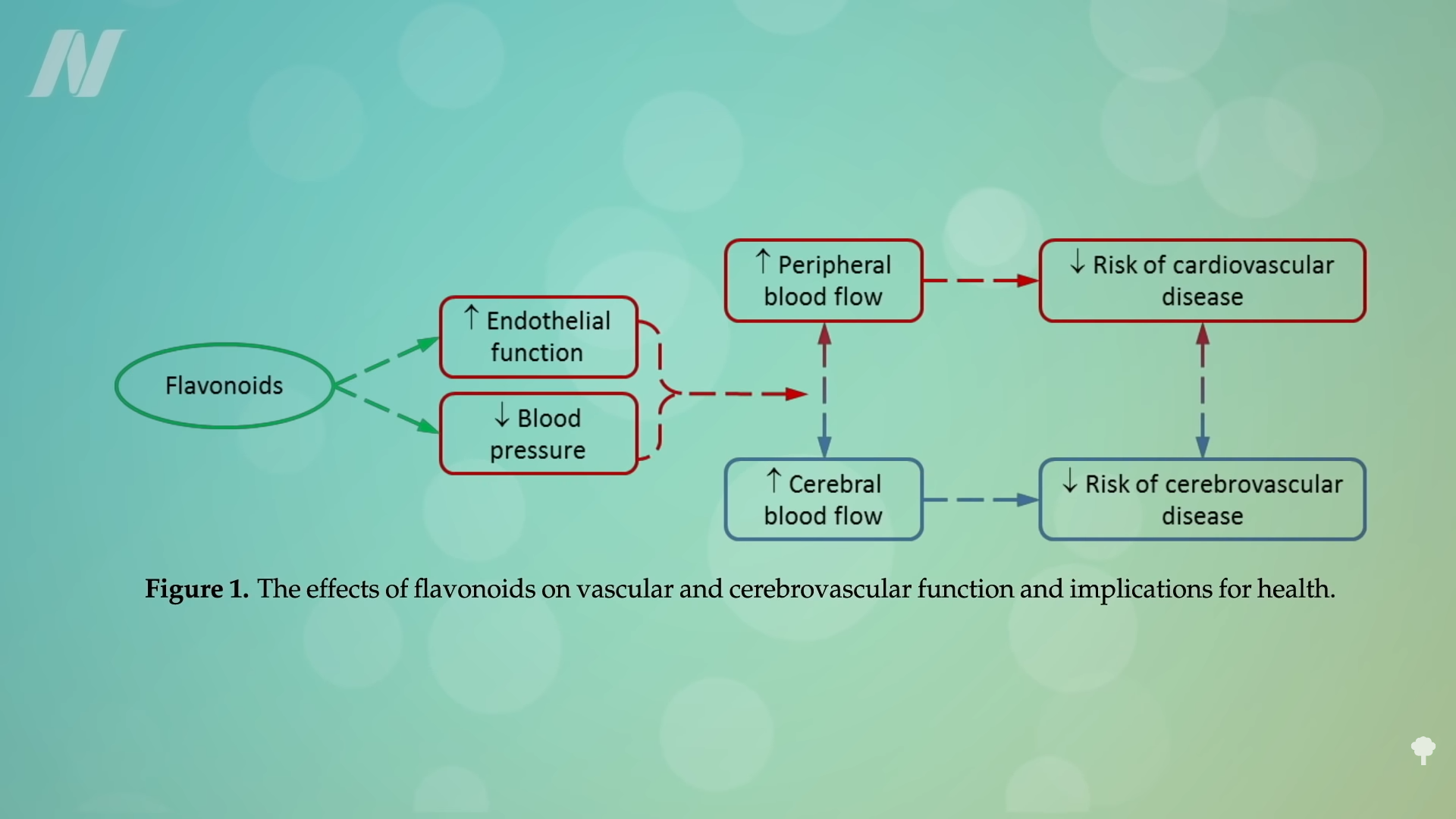
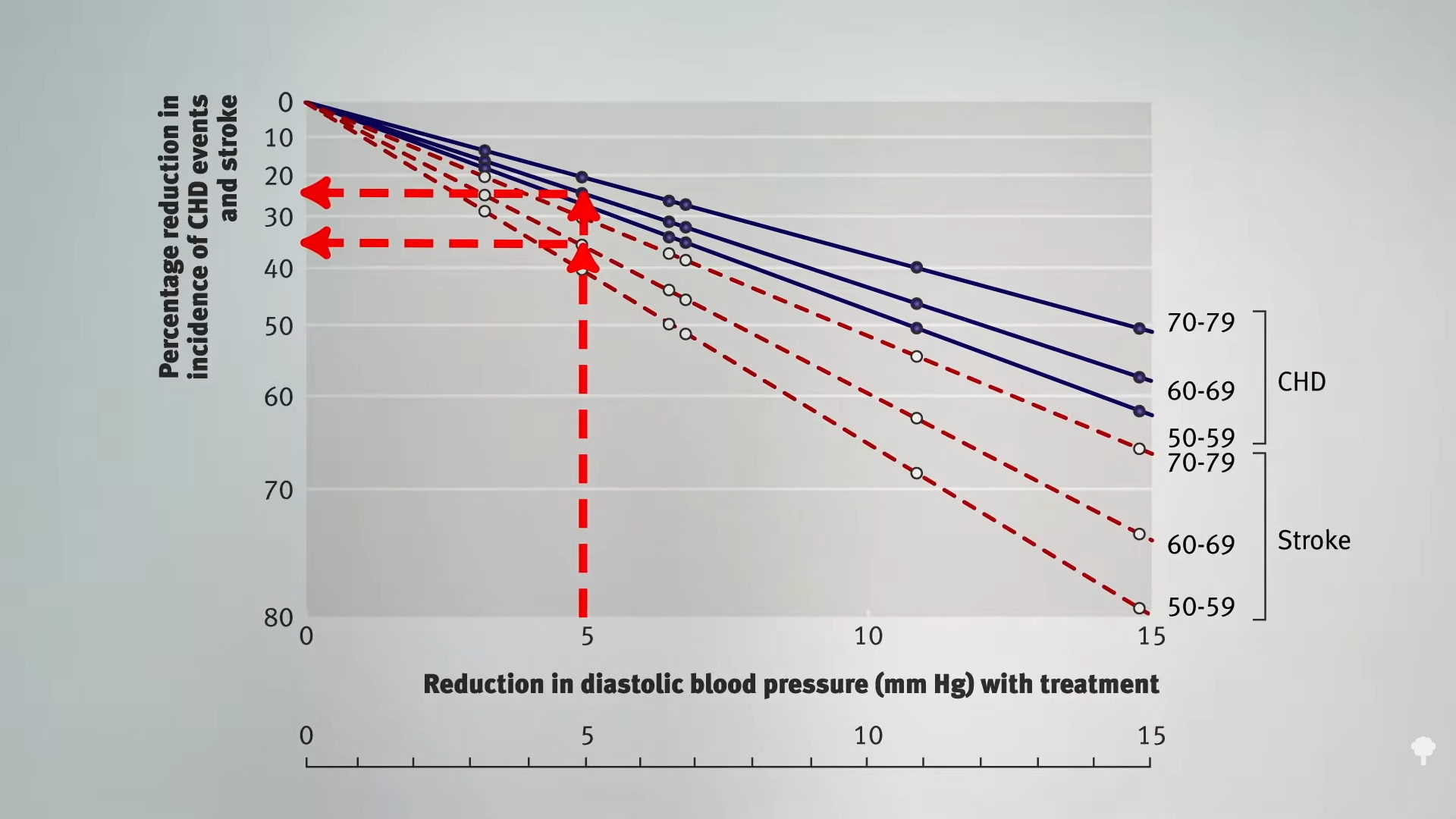
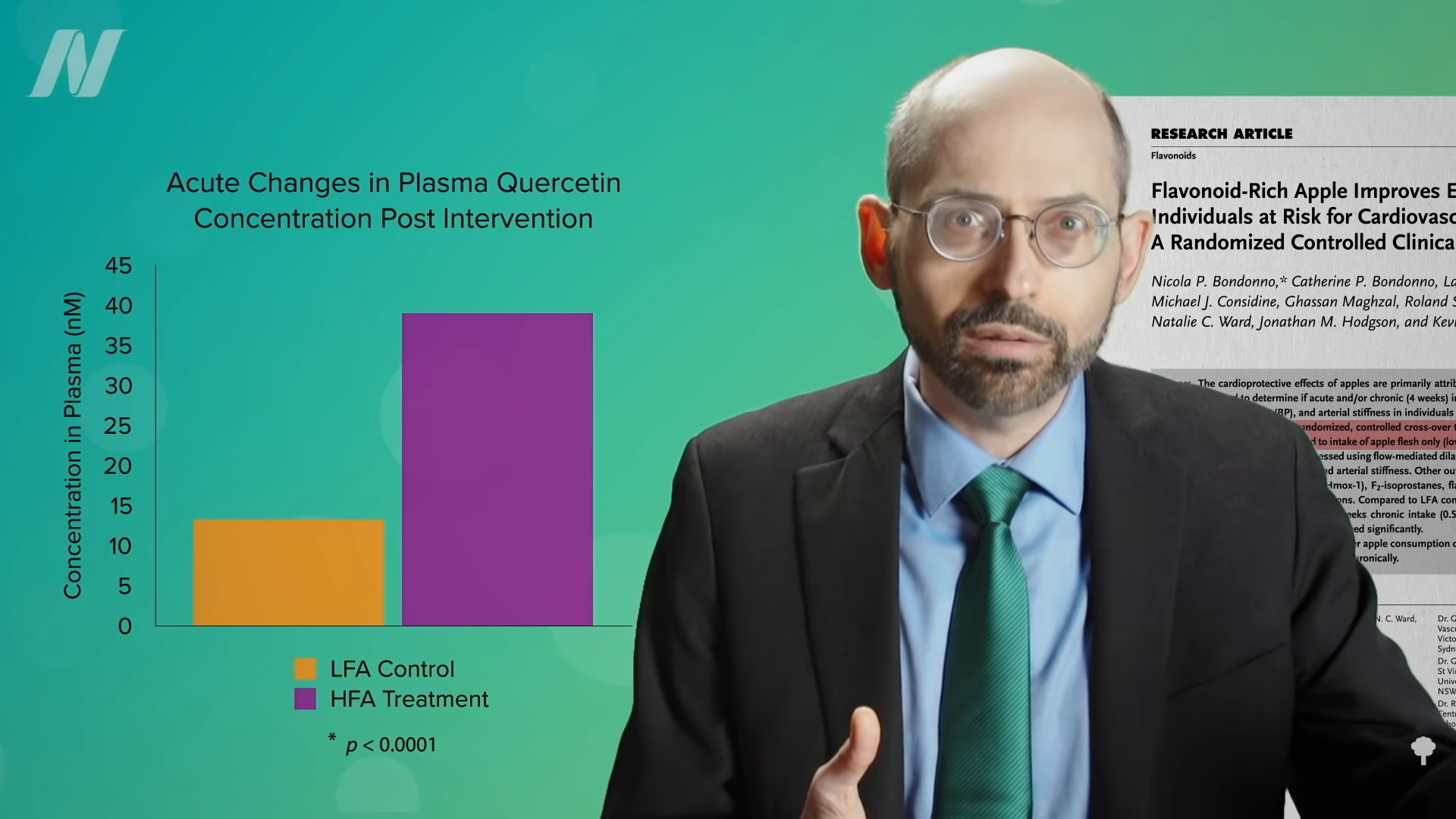 Even compared to spinach? As you can see in the graph below and at 3:14 in my
Even compared to spinach? As you can see in the graph below and at 3:14 in my  What’s nice about these results is that we’re talking about whole foods, not some supplement or extract. So, easily, “this could be translated into a natural and low-cost method of reducing the cardiovascular risk profile of the general population.”
What’s nice about these results is that we’re talking about whole foods, not some supplement or extract. So, easily, “this could be translated into a natural and low-cost method of reducing the cardiovascular risk profile of the general population.” 






