This cross-section of a rat brain shows tissue from a human brain organoid fluorescing in light green. Scientists say these implanted clusters of human neurons could aid the study of brain disorders.
Pasca lab / Stanford Medicine
hide caption
toggle caption
Pasca lab / Stanford Medicine
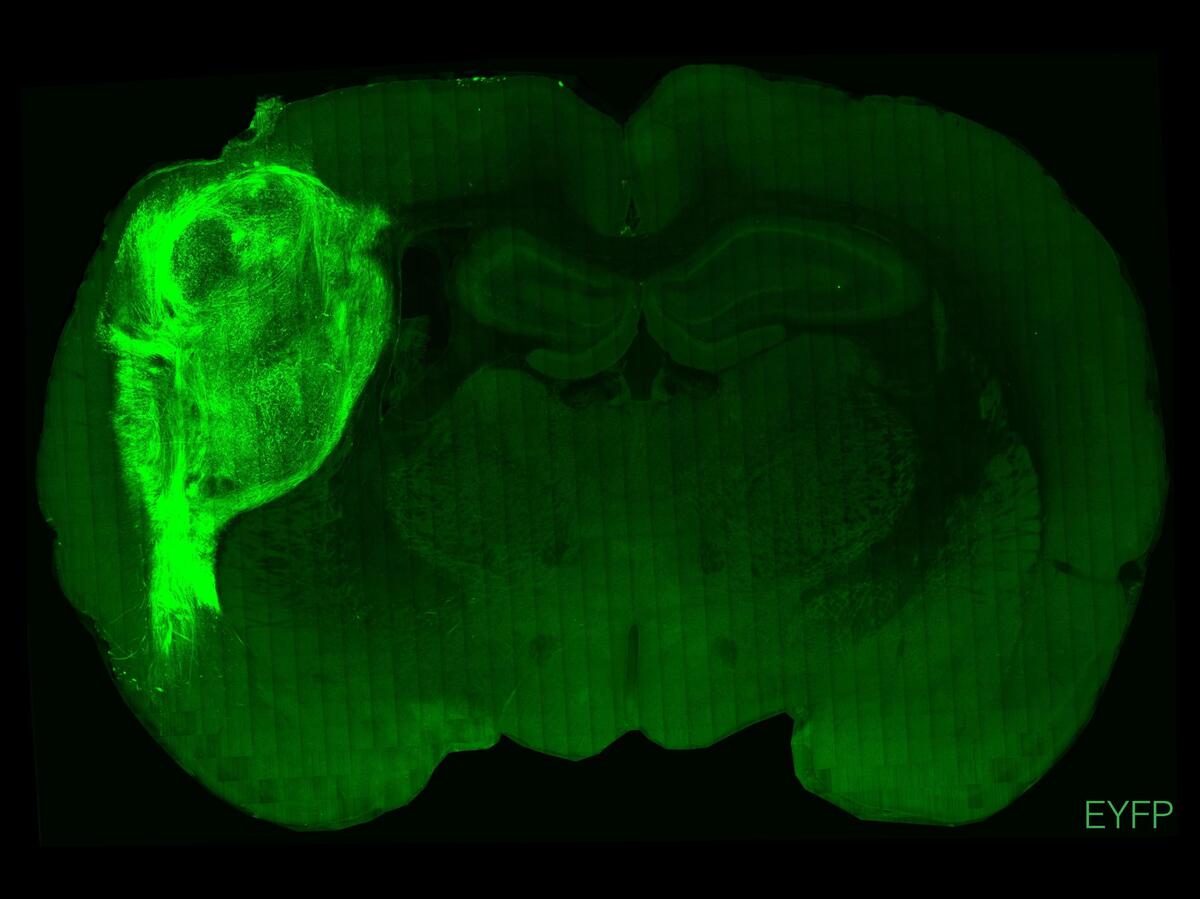
This cross-section of a rat brain shows tissue from a human brain organoid fluorescing in light green. Scientists say these implanted clusters of human neurons could aid the study of brain disorders.
Pasca lab / Stanford Medicine
Scientists have demonstrated a new way to study conditions like autism spectrum disorder, ADHD, and schizophrenia.
The approach involves transplanting a cluster of living human brain cells from a dish in the lab to the brain of a newborn rat, a team from Stanford University reports in the journal Nature.
The cluster, known as a brain organoid, then continues to develop in ways that mimic a human brain and may allow scientists to see what goes wrong in a range of neuropsychiatric disorders.
“It’s definitely a step forward,” says Paola Arlotta, a prominent brain organoid researcher at Harvard University who was not involved in the study. “The ultimate goal of this work is to begin to understand features of complex diseases like schizophrenia, autism spectrum disorder, bipolar disorder.”
But the advance is likely to make some people uneasy, says bioethicist Insoo Hyun, director of life sciences at the Museum of Science in Boston and a member of the Harvard Medical School Center for Bioethics.
“There is a tendency for people to assume that when you transfer the biomaterials from one species into another, you transfer the essence of that animal into the other,” Hyun says, adding that even the most advanced brain organoids are still very rudimentary versions of a human brain.
Clearing a scientific obstacle
The success in transplanting human brain organoids into a living animal appears to remove a major barrier to using them as models of human disease. It also represents the culmination of seven years of work overseen by Dr. Sergiu Pasca, a professor of psychiatry and behavioral sciences at Stanford.
Human brain organoids are made from pluripotent stem cells, which can be coaxed into becoming various types of brain cells. These cells are grown in a rotating container known as a bioreactor, which allows the cells to spontaneously form brain-like spheres about the size of a small pea.
But after a few months, the lab-grown organoids stop developing, says Pasca, whose lab at Stanford devised the transplant technique. Individual neurons in the cluster remain relatively small, he says, and make relatively few connections.
“No matter how long we keep them in a dish, they still do not become as complex as human neurons would be in an actual human brain,” Pasca says. That may be one reason organoids have yet to reveal much about the origins of complex neuropsychiatric disorders, he says.
So Pasca’s team set out to find an environment for the organoids that would allow them to continue growing and maturing. They found one in the brains of newborn rats.
“We discovered that the [organoid] grows, over the span of a few months, about nine times in volume,” Pasca says. “In the end it covers roughly about a third of a rat’s hemisphere.”
The transplanted cells don’t seem to cause problems for the rats, who behave normally as they grow, Pasca says.
“The rat tissue is just pushed aside,” he says. “But now you also have a group of human cells that are integrating into the circuitry.”
The human cells begin to make connections with rat cells. Meanwhile, the rat’s blood vessels begin to supply the human cells with oxygen and nutrients.
A link to the senses
Pasca’s team placed each organoid in an area of the rat brain that processes sensory information. After a few months, the team did an experiment that suggested the human cells were reacting to whatever the rat was sensing.
“When you stimulate the whiskers of the rat, the majority of human neurons are engaged in an electrical activity that follows that stimulation,” Pasca says.
Another experiment suggests the human cells could even influence a rat’s behavior.
The team trained rats to associate stimulation of their human cells with a reward – a drink of water. Eventually, the rats began to seek water whenever the human cells were stimulated.
In a final experiment, Pasca’s team set out to show how transplanted organoids could help identify the brain changes associated with a specific human disorder. They chose Timothy Syndrome, a very rare genetic disorder that affects brain development in ways that can cause symptoms of autism spectrum disorder.
The team compared organoids made from the stem cells of healthy people with organoids made from the stem cells of patients with the syndrome. In the lab, the cell clusters looked the same.
“But once we transplanted and we looked 250 days later, we discovered that while control cells grew dramatically, patient cells failed to do so,” Pasca says.
A better model, with ethical concerns
The experiments show that Pasca’s team has developed a better model for studying human brain disorders, Arlotta says.
The key seems to be providing the transplanted organoids with sensory information that they don’t get growing in a dish, she says, noting that an infant’s brain needs this sort of stimulation to develop normally.
“It’s the stuff that we get after we are born,” she says, “especially when we begin to experience the world and hear sound, see light, and so on.”
But as brain organoids become more like actual human brains, scientists will have to consider the ethical and societal implications of this research, Arlotta says.
“We need to be able to watch it, consider it, discuss it and stop it if we think we think one day we are at the point where we shouldn’t progress,” she says. “I think we are far, far away from that point right now.”
Even the most advanced brain organoids have nothing even remotely like the capabilities of a human brain, says Hyun, who posted a video conversation he had with Pasca to coincide with the publication of the new study.
Yet many ethical discussions have focused on the possibility that an organoid could attain human-like consciousness.
“I think that’s a mistake,” Hyun says. “We don’t exactly know what we mean by ‘human-like consciousness,’ and the nearer issue, the more important issue, is the well-being of the animals used in the research.”
He says that wasn’t a problem in the Pasca lab’s experiments because the organoids didn’t seem to harm the animals or change their behavior.
If human brain organoids are grown in larger, more complex animal brains, Hyun says, the cell clusters might develop in ways that cause the animals to suffer.
“What I’m concerned about,” he says, “is what’s next.”
Jon Hamilton
Source link









